10 Essential Skills for Drug Safety Jobs in 2025

Introduction
The landscape of drug safety is undergoing a significant transformation, propelled by escalating regulatory demands and the imperative for enhanced patient protection. With the global market for drug safety monitoring poised for substantial growth, professionals in this arena must cultivate a diverse skill set to adeptly navigate the complexities of pharmacovigilance.
What essential skills will delineate success in drug safety roles by 2025? How can professionals position themselves advantageously in this dynamic environment? This article explores ten critical competencies that will define the future of drug safety positions, ensuring that practitioners are thoroughly prepared to tackle the challenges and seize the opportunities that lie ahead.
AVS Life Sciences: Expertise in Drug Safety and Pharmacovigilance
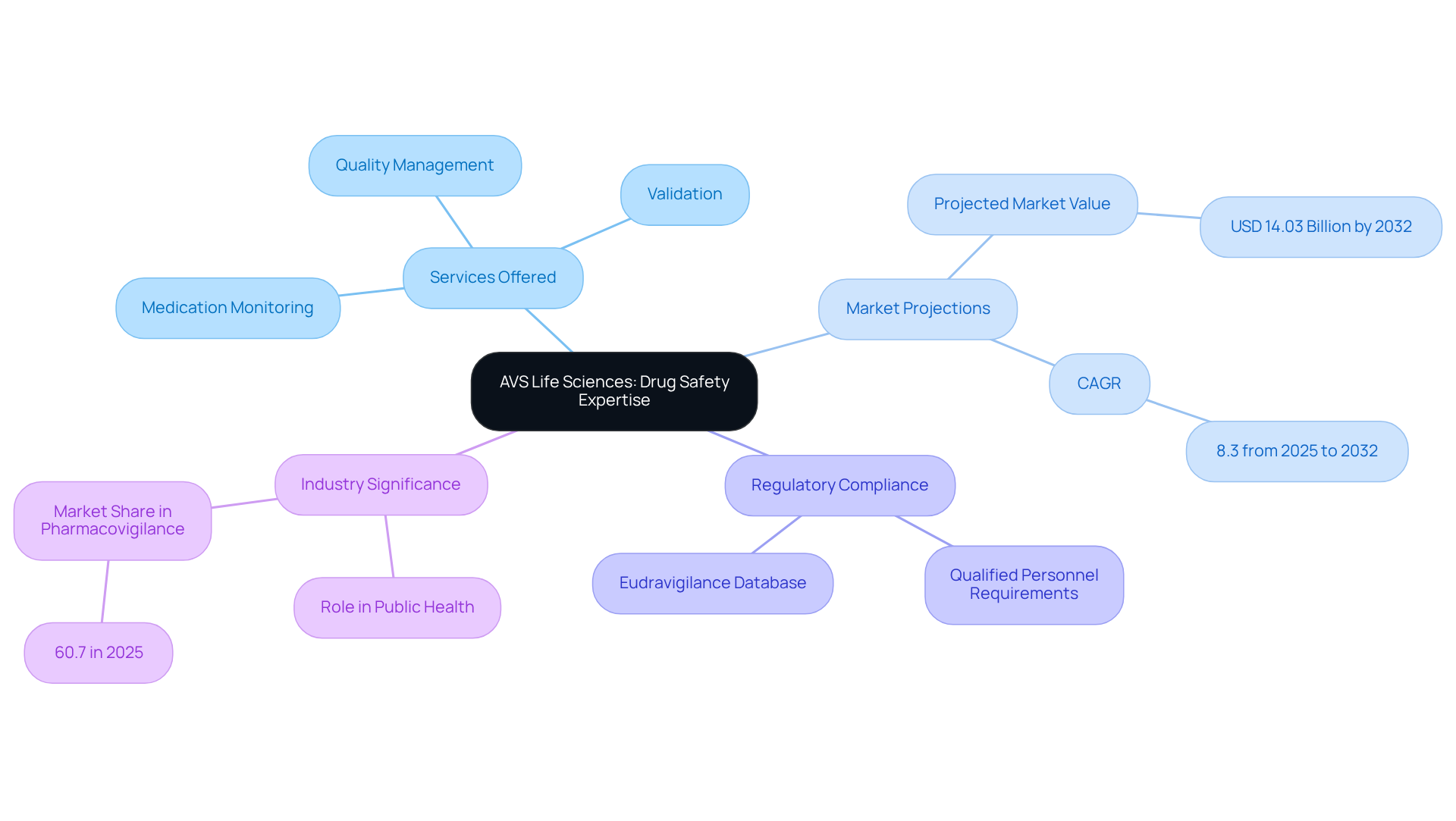
AVS Life Sciences stands at the forefront of and tailored for the pharmaceutical and biotechnology sectors. Their comprehensive offerings include validation, , and —essential components for navigating the intricate regulatory landscapes governing pharmaceutical development.
With a , AVS Life Sciences is unwavering in its commitment to maintaining the and compliance throughout the entire lifecycle of medicinal products. This dedication is particularly crucial as the global drug safety monitoring market is projected to reach USD 14.03 billion by 2032, reflecting a compound annual growth rate (CAGR) of 8.3% from 2025 to 2032.
The firm’s expertise not only ensures adherence to stringent regulations but also positions them as a trusted partner for organizations aiming to implement effective medication monitoring programs. As oversight intensifies, the significance of in escalates, underscoring the vital role AVS Life Sciences plays in safeguarding public health and safety.
Moreover, with national competent authorities and the European Medicines Agency (EMA) mandating qualified personnel, AVS Life Sciences is exceptionally equipped to fulfill these requirements. Their extensive industry expertise, which includes GMP audits across , contract testing laboratories, and manufacturing locations, fortifies their strategic positioning in the market, projected to hold the largest share of 60.7% in 2025.
Additionally, the is instrumental in disseminating drug safety information among competent authorities, further emphasizing the significance of AVS's services in ensuring . Monitoring unique circumstances, such as pregnancy and pediatric groups, is also a critical element of effective drug safety practices that AVS Life Sciences adeptly addresses.
As the market evolves, the responsibilities of marketing authorization holders (MAHs) in to shape the landscape, making AVS's expertise indispensable.
Regulatory Compliance: Mastering Industry Standards and Guidelines
A thorough understanding of compliance with regulations is essential for professionals in , particularly concerning , , and . Mastery of these frameworks is crucial for ensuring that all adhere to legal and ethical standards, ultimately protecting public health. in are imperative as they enable professionals to stay abreast of evolving regulations and maintain compliance effectively.
The pharmaceutical industry faces significant , which underscores the need for , as evidenced by the FDA issuing 3,344 observations for noncompliance in 2018 alone. Firms that excel in mastering GMP and ISO standards, such as AVS Life Sciences, exemplify the importance of robust quality management systems for that not only meet compliance requirements but also enhance operational efficiency. AVS Life Sciences provides extensive consulting services that assist organizations in navigating the complexities of compliance and quality management related to , ensuring adherence to the highest standards.
As policy modifications continue to influence the landscape of [drug safety jobs](https://avslifesciences.com/blog-post/10-essential-steps-for-gdufa-self-identification-compliance) in 2025, experts must adapt to new benchmarks and methodologies. The adoption rates of ISO standards in pharmaceutical quality are steadily increasing, reflecting a growing acknowledgment of their significance in ensuring product quality and safety. By fostering a culture of adherence and continuous improvement, organizations can more effectively manage the complexities of regulatory landscapes and ensure the quality and effectiveness of drug safety jobs.

Risk Management: Identifying and Mitigating Safety Concerns
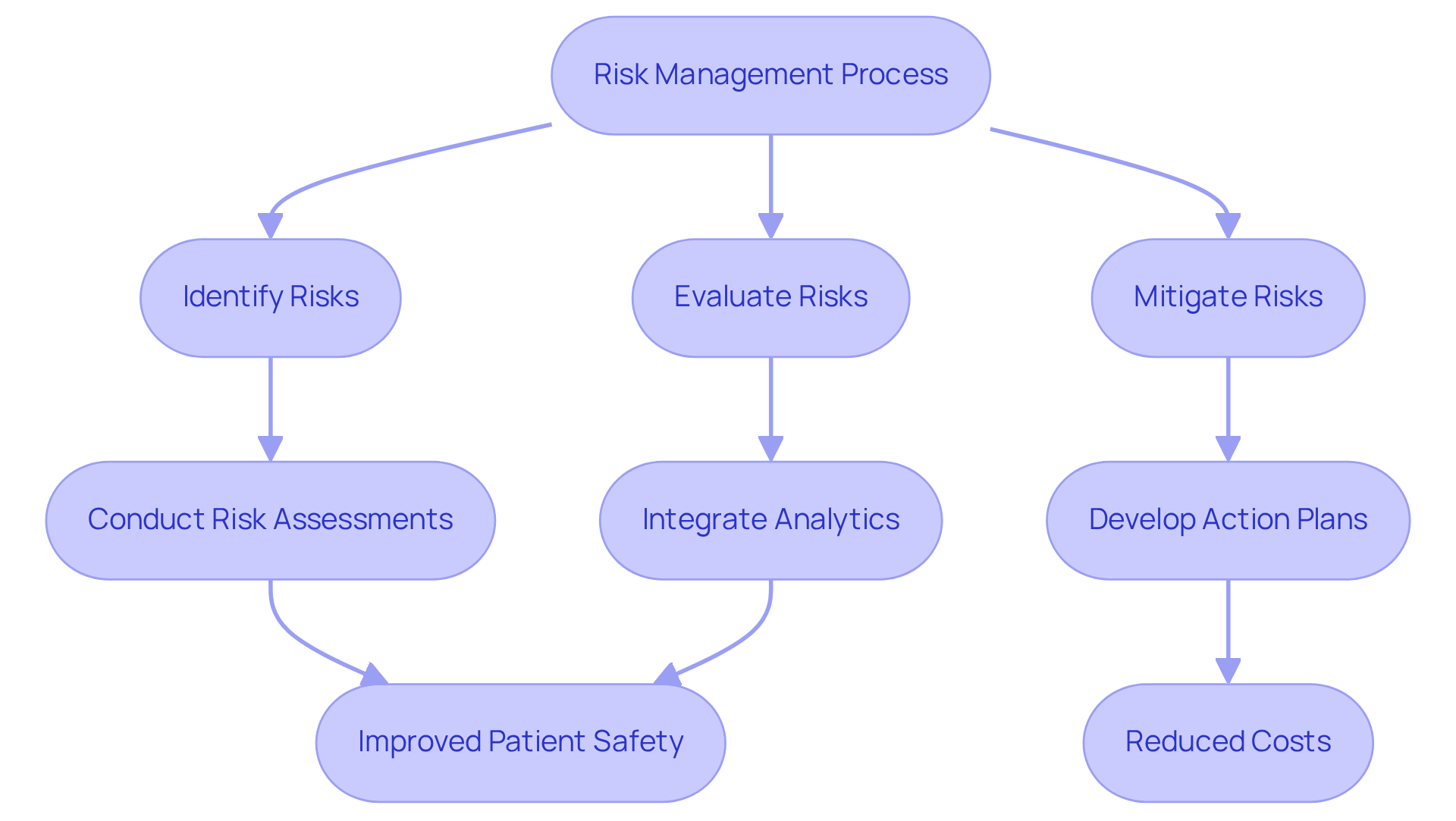
represents a systematic approach that involves identifying, evaluating, and mitigating potential health risks associated with medicinal products. Professionals in this field must excel in , which are vital for recognizing adverse impacts and safeguarding throughout the drug lifecycle. Effective risk management plans are essential; they outline specific actions to address identified risks and integrate proactive strategies to minimize adverse effects.
Recent trends indicate a significant shift towards . For instance, the application of risk analysis has led to an impressive 86% reduction in negative event reporting durations, decreasing from an average of 16.2 days to just 2.3 days. Moreover, healthcare systems that have adopted extensive analytics programs have experienced a 23% reduction in costs associated with preventable adverse events, resulting in a remarkable 430% .
Case studies from 2025 underscore the effectiveness of . A notable example is a , which reported a 42% reduction in medication errors, preventing approximately 1.2 million errors annually. This illustrates the importance of integrating and fostering a culture that prioritizes .
As the pharmaceutical landscape continues to evolve, staying informed about these trends and employing robust will be imperative for professionals aiming to enhance drug security and efficacy. AVS Life Sciences provides comprehensive that can bolster these initiatives, particularly in overcoming challenges such as data integration across disparate systems. By leveraging AVS Life Sciences' expertise, professionals can fortify their and ultimately improve .
Data Analysis: Interpreting Safety Data for Informed Decisions
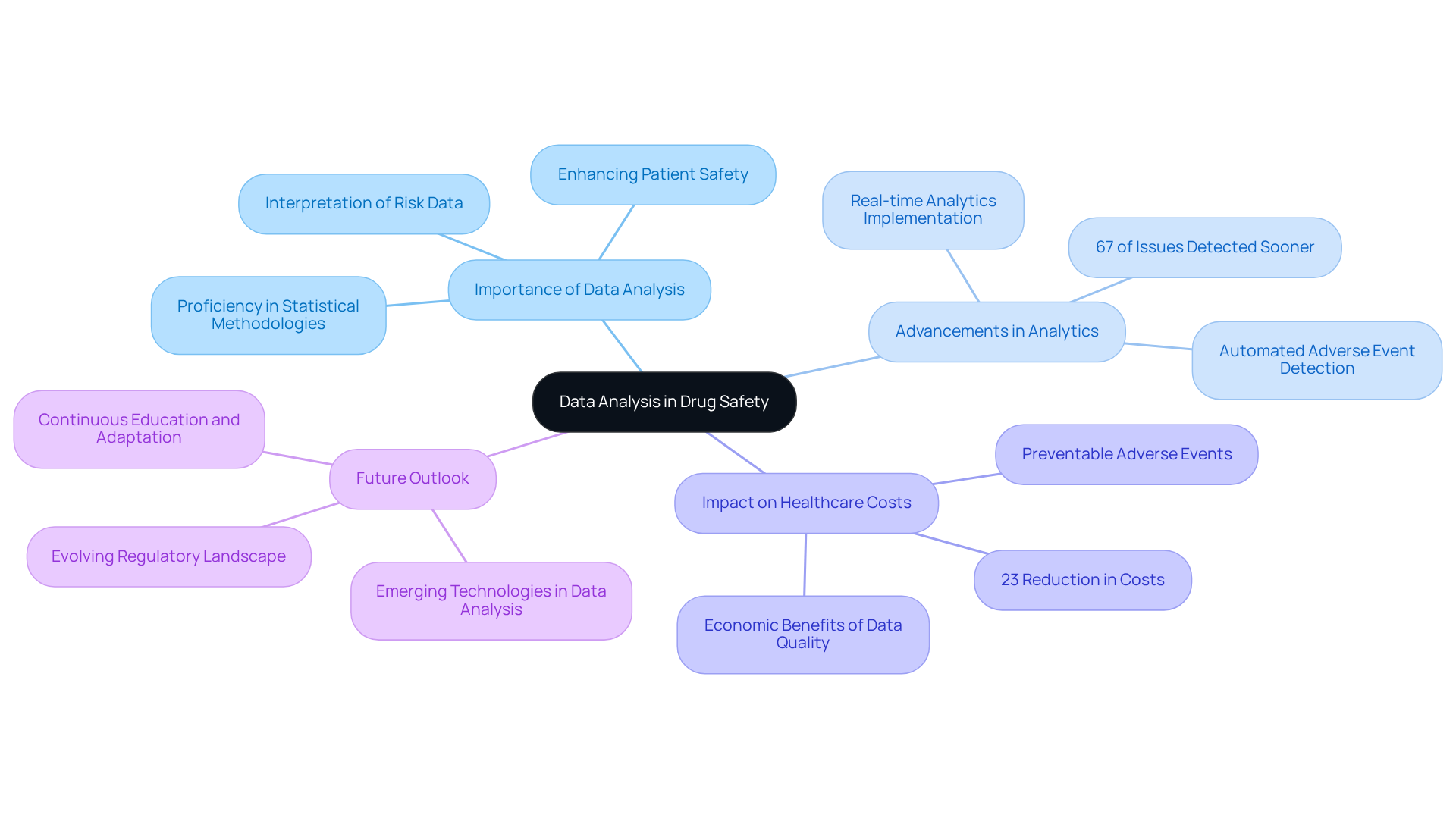
Data analysis stands as a critical competency for , enabling precise interpretation of risk data. This involves a thorough examination of adverse event reports, identifying patterns, and evaluating the . Proficiency in statistical methodologies and is essential for extracting actionable insights from intricate datasets, ultimately guiding compliance decisions and enhancing patient safety.
Recent advancements in for risk management have significantly bolstered the ability to detect signals earlier. Studies reveal that 67% of serious issues could be identified an average of 18 months sooner using advanced analytics. This but also cultivates a culture of well-being within healthcare organizations, benefiting professionals in . Moreover, entities that emphasize data quality can realize a 23% reduction in healthcare costs linked to preventable adverse events.
As Albert Einstein astutely noted, "Concern for humanity and his well-being must always constitute the primary focus of all technical efforts." This sentiment resonates deeply within the realm of , where is vital for safeguarding public health. Additionally, as Terry J. Moulton articulated, "A ," underscoring the ongoing commitment required in .
Looking ahead to 2025, the landscape of pharmaceutical oversight will continue to evolve, with an increasing emphasis on leveraging to refine decision-making processes. As specialists in the field advocate for continuous education and adaptation to emerging technologies, the ability to analyze health data effectively will remain a cornerstone of successful . To maintain a competitive edge, professionals should engage in regular training on the latest and methodologies.
Communication Skills: Collaborating Across Teams and Stakeholders
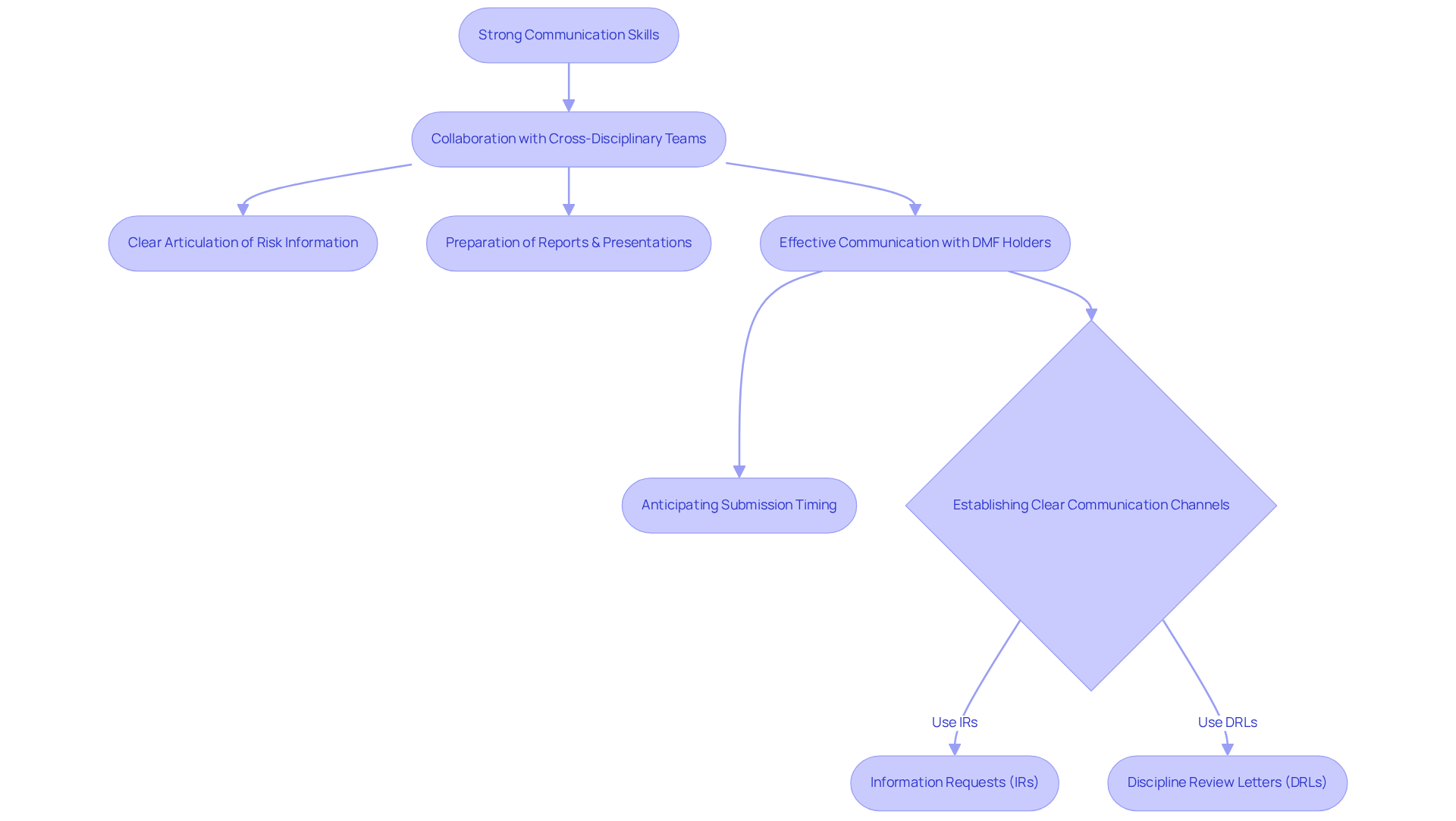
Strong communication abilities are essential for , who frequently collaborate with cross-disciplinary teams, regulatory bodies, and healthcare providers. The capacity to articulate intricate risk information clearly and concisely is vital for ensuring that all stakeholders comprehend potential hazards and precautionary measures. This encompasses the preparation of , the delivery of informative presentations, and participation in discussions that foster transparency and collaboration in pharmaceutical initiatives.
In 2025, the significance of is underscored by the necessity for among team members and stakeholders. For instance, with is crucial to anticipate submission timing, as delays can adversely affect ANDA goal dates. Furthermore, establishing clear communication channels, such as Information Requests (IRs) and Discipline Review Letters (DRLs), facilitates the exchange of critical information, ensuring that necessary data is submitted promptly.
emphasize cooperation, which not only enhances the quality of evaluations but also cultivates trust among team members. By promoting a culture of , professionals can exchange insights and proactively address issues, ultimately leading to improved patient care outcomes. As the field of pharmacovigilance evolves, mastering skills will remain a fundamental element for success in drug safety jobs related to medication monitoring. Combining these skills with can further augment the effectiveness of pharmaceutical protection initiatives.
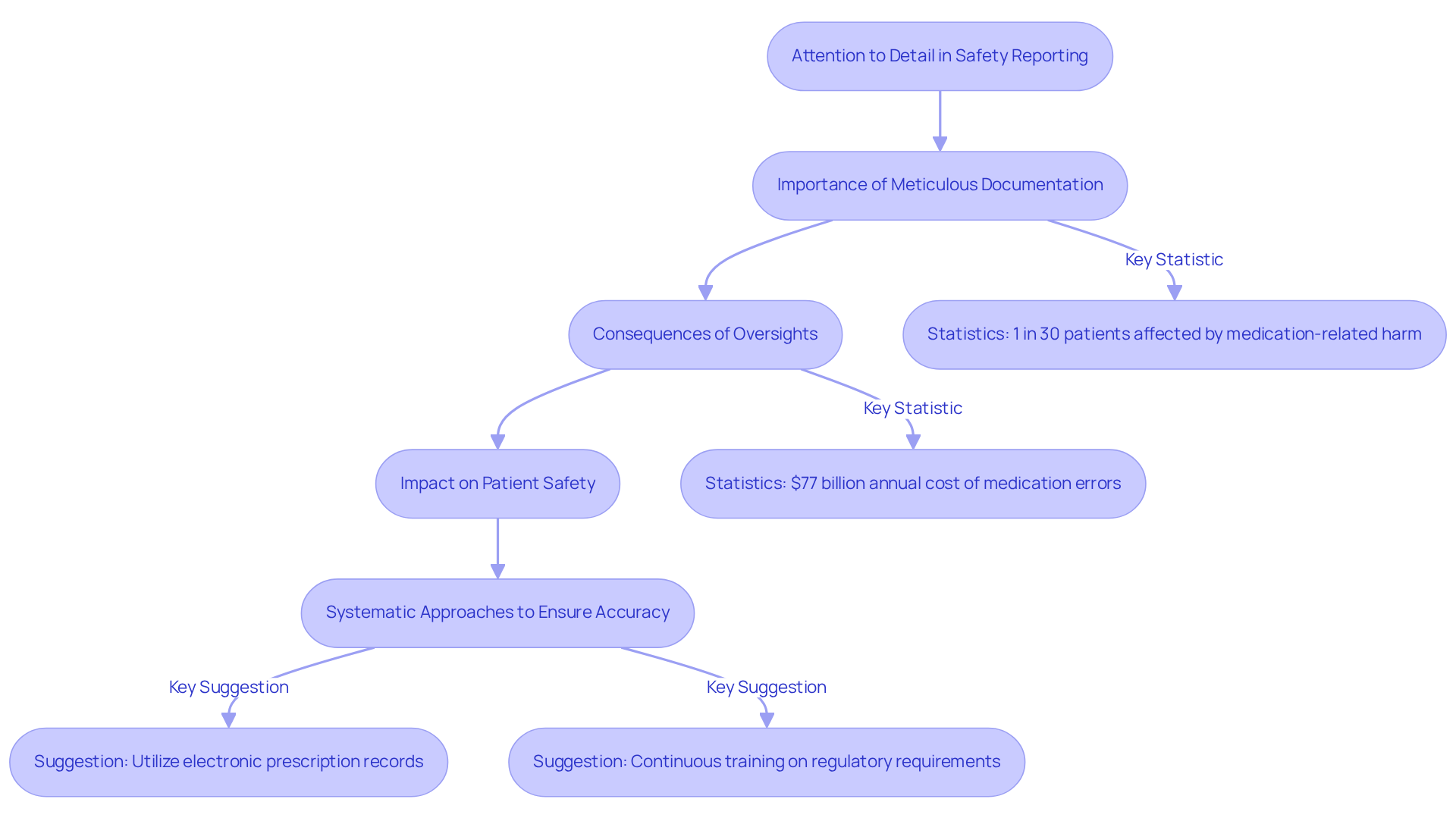
Attention to Detail: Ensuring Accuracy in Safety Reporting
is a crucial ability for professionals in drug safety jobs, significantly affecting the precision of reporting on risk. Meticulous documentation of is essential, as it ensures that all pertinent information is captured and reported in accordance with . This diligence is vital in preventing mistakes that could endanger patient well-being and uphold the integrity of the .
Recent audits in 2025 have underscored the significance of precision in , revealing that even minor oversights can lead to substantial consequences. For instance, medication-related harm affects 1 out of every 30 patients, with over a quarter of these cases being severe or life-threatening. Such statistics highlight the critical need for professionals to focus on precision in their reporting practices.
Experts emphasize that not only enhances compliance but also directly impacts . A culture of thoroughness in documentation can significantly reduce the incidence of , which are estimated to cost the healthcare system $77 billion annually. By establishing strong and promoting a culture where precision is valued, organizations can mitigate risks related to drug safety jobs.
To ensure , professionals should adopt systematic approaches, such as utilizing electronic prescription records and medication bar coding, which help safeguard against errors. Furthermore, on the latest regulatory requirements and best practices are crucial for maintaining high standards in reporting.
In conclusion, the commitment to detail in reporting on security is not merely a procedural necessity; it is a fundamental aspect of protecting patient health and enhancing the overall efficacy of the healthcare system.
Project Management: Overseeing Drug Safety Initiatives
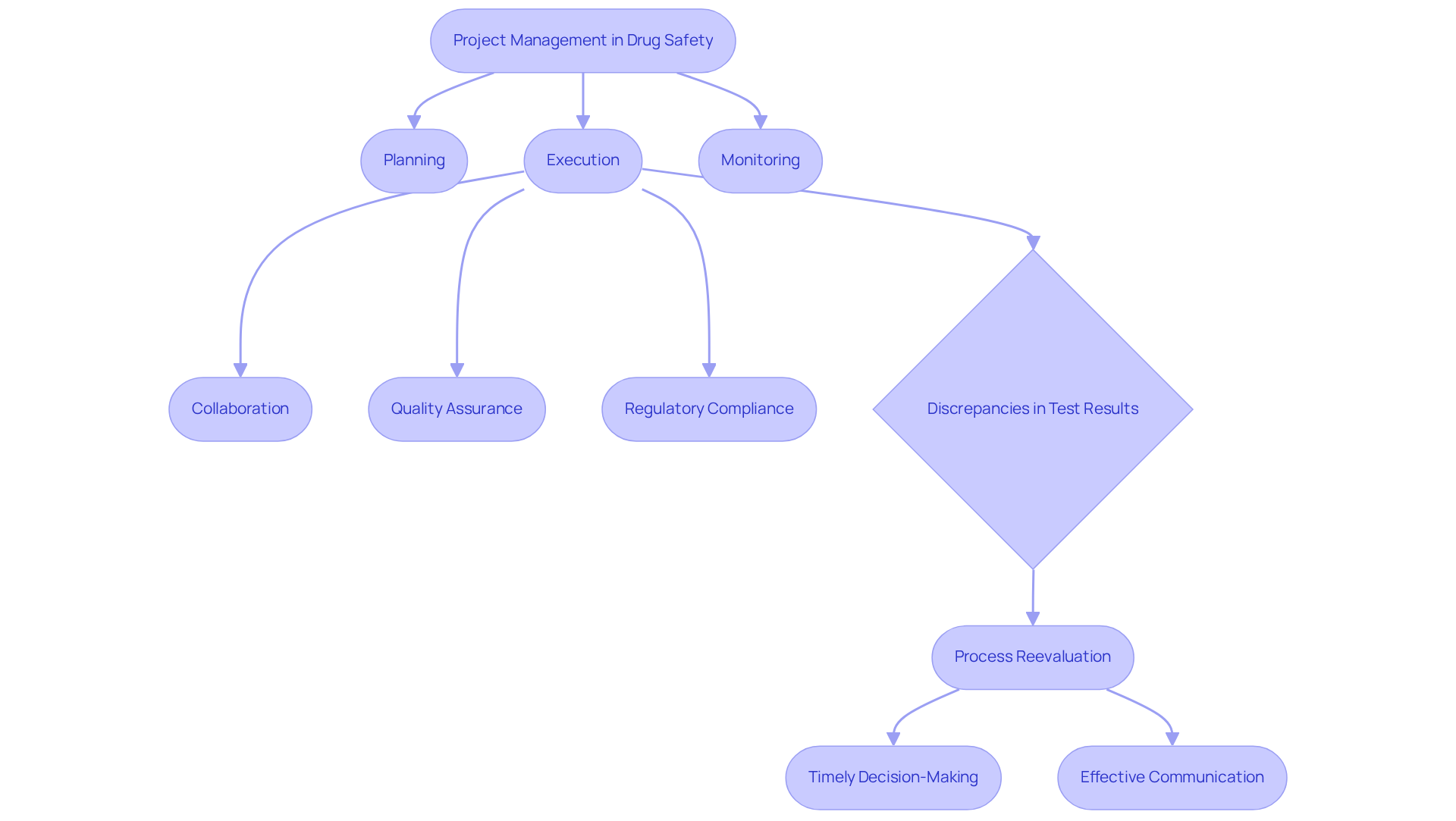
stands as a pivotal skill for those pursuing drug safety jobs, particularly within the realm of pharmacovigilance. This discipline encompasses the planning, execution, and monitoring of safety evaluations, ensuring that all activities align with legal timelines and budget constraints. Effective fosters collaboration among team members, a crucial component for maintaining adherence to stringent legal requirements.
A exemplifies this principle, showcasing their . This project adhered not only to budget and timeline but also underscored the importance of , highlighting the critical role of in safeguarding data integrity.
During the upgrade, challenges emerged, including discrepancies in test results attributed to improperly installed barcode scanner cameras, which were initially overlooked. This experience yielded valuable lessons, prompting the QC laboratory team and to reassess their processes and enhance the reliability of test results.
As the demand for skilled escalates, professionals must adeptly navigate complex regulatory landscapes and leverage advanced tools. Insights from industry experts emphasize that are vital for the successful implementation of pharmaceutical initiatives.
As the industry evolves, the integration of innovative practices will be essential for bolstering the reliability and efficacy of pharmaceuticals.
Pharmacovigilance Systems: Monitoring Adverse Drug Reactions
are crucial for tracking and ensuring the security of treatments. As we look towards 2025, advancements in these systems have led to more efficient . Experts must be well-versed in the various systems employed to gather, examine, and present data on safety, including and databases. Understanding how to utilize these systems effectively empowers professionals in to recognize hazard signals and respond promptly to emerging issues.
Current trends indicate a significant shift towards the integration of technology in , with becoming increasingly prevalent. Statistics reveal that serious ADRs accounted for 34.1% of total reports, with a mortality rate of 2.8% among serious ADRs, underscoring the necessity for robust . Efficient electronic reporting instruments, such as the Eudravigilance database, play a crucial role in exchanging among competent authorities, ensuring that potential risk factors are communicated effectively.
Furthermore, the continuous development of drug safety emphasizes the importance of educating healthcare professionals about and the use of these tools. By enhancing their skills in ADR monitoring, professionals can contribute to improved patient care and adherence to regulatory standards, including the submission of . As the landscape of medication security continues to evolve, remaining informed about the latest advancements and trends in is essential for success in the field.

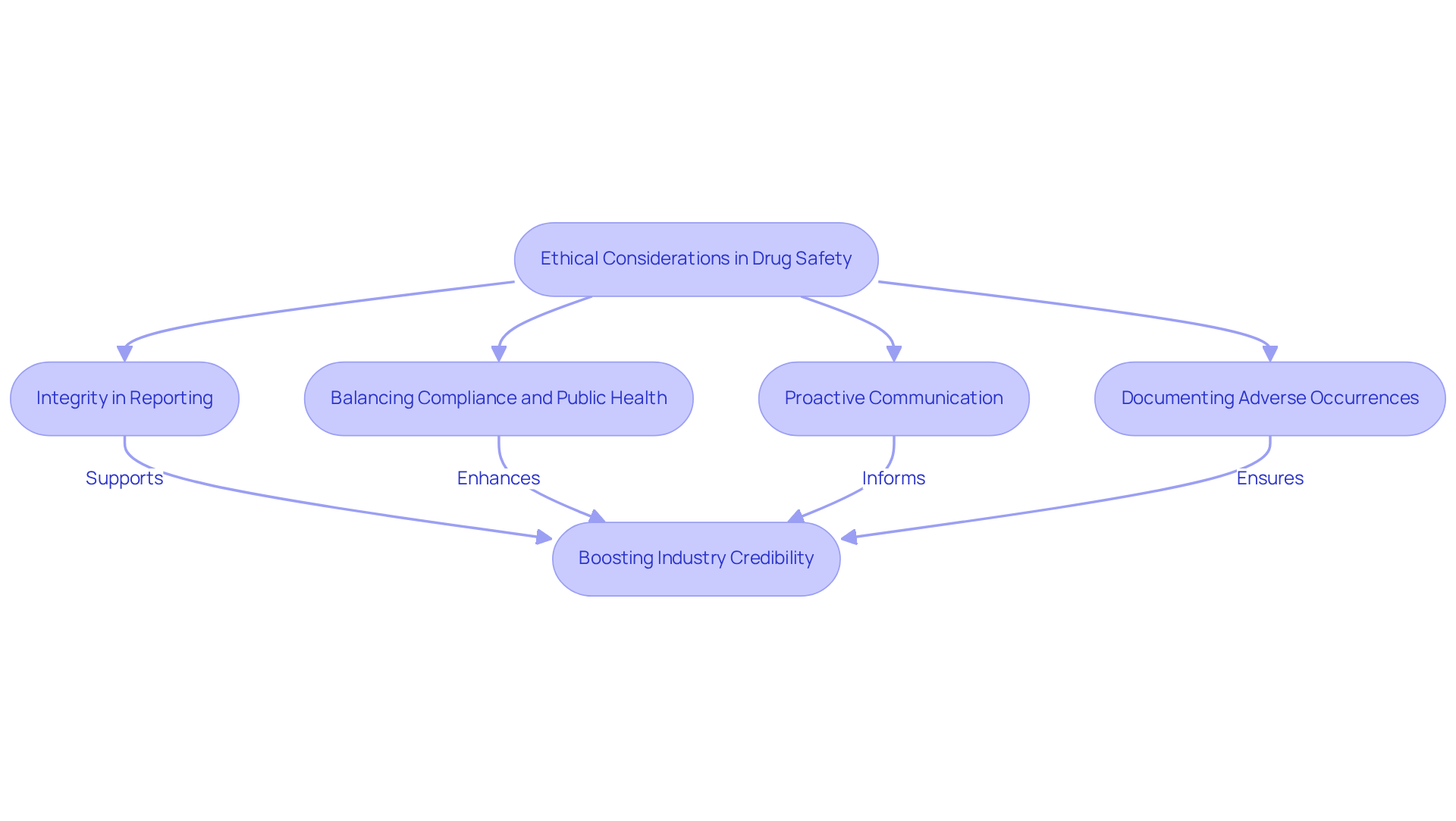
Ethical Considerations: Upholding Integrity in Drug Safety
, serving as the foundation for decision-making among professionals in the field. Maintaining integrity necessitates precise and transparent reporting of all protection data, with a firm dedication to prioritizing patient welfare. Substance security specialists confront complex , where they must balance and cultivate trust in the pharmaceutical sector.
Recent have highlighted the necessity for robust , as lapses can lead to significant repercussions for both patients and companies. and proactive communication with regulatory agencies illustrate integrity in reporting risk data, ensuring that all stakeholders are informed and involved.
By upholding high ethical standards, pharmaceutical professionals not only safeguard patients but also as a whole.
Continuous Learning: Adapting to Evolving Industry Standards
Ongoing education stands as a cornerstone for pharmaceutical professionals, particularly as the industry undergoes relentless transformation driven by new regulations, technologies, and best practices. Engaging in —such as attending workshops and participating in —empowers professionals in to remain abreast of the and pharmacovigilance. This dedication not only sharpens individual expertise but also significantly contributes to by ensuring the safety and efficacy of pharmaceutical products.
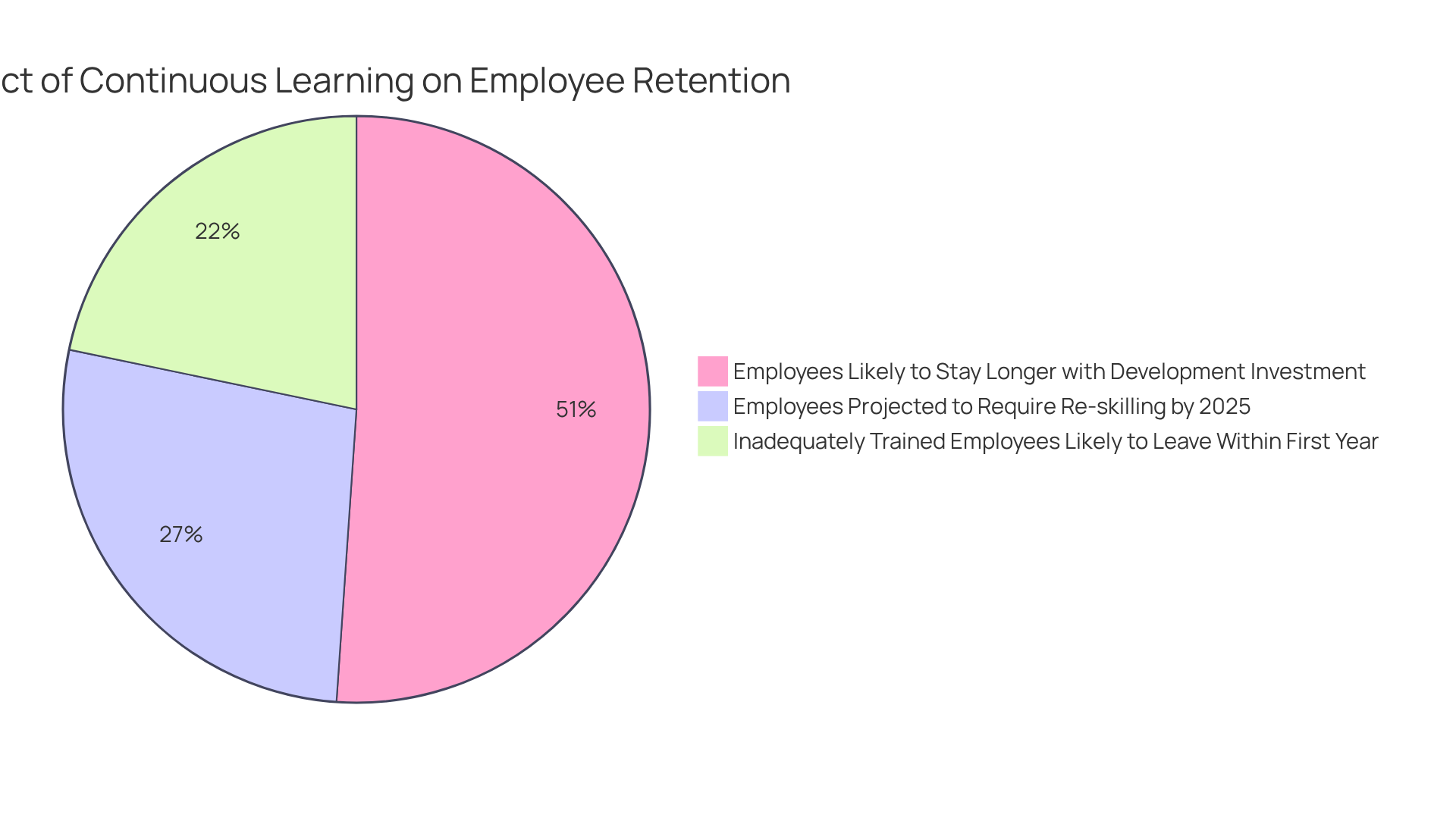
Notably, statistics indicate that:
- 94% of employees are likely to stay longer at a company that invests in their learning and development, highlighting the .
- Projections suggest that 50% of employees will require re-skilling by 2025, the urgency for effective learning strategies has never been more pronounced.
- within the first year, underscoring the repercussions of insufficient training.
As the at a compound annual growth rate of 15% until 2027, it is imperative for professionals to prioritize their growth to adeptly and maintain compliance. Albert Einstein aptly stated, "Once you stop learning, you start dying," which underscores the .
Conclusion
The landscape of drug safety jobs in 2025 presents a complex interplay of regulatory compliance, risk management, data analysis, and ethical considerations. As the industry evolves, the essential skills required to excel in this field are increasingly specialized. Professionals must not only grasp the intricacies of pharmacovigilance but also master the tools and methodologies that underpin effective drug safety practices.
Key insights discussed throughout this article underscore the critical importance of communication, attention to detail, and project management in ensuring the safety and efficacy of pharmaceutical products. The role of AVS Life Sciences stands out as a pivotal player, providing the expertise and support necessary to navigate the regulatory landscape and enhance drug safety initiatives. Continuous learning and adaptation to emerging technologies are vital components for success, ensuring professionals remain equipped to meet the demands of an ever-changing environment.
In summary, the future of drug safety jobs is contingent upon a steadfast commitment to excellence, collaboration, and ethical integrity. As the industry confronts new challenges and opportunities, professionals are urged to embrace ongoing education and proactive engagement with the latest advancements. By fostering a culture of continuous improvement and adhering to high standards, the pharmaceutical sector can enhance patient safety and uphold public trust, ultimately contributing to a healthier society.
Frequently Asked Questions
What services does AVS Life Sciences provide?
AVS Life Sciences offers drug safety jobs and medication monitoring services, including validation, regulatory compliance, and quality management for the pharmaceutical and biotechnology sectors.
How many professionals work at AVS Life Sciences?
AVS Life Sciences has a dedicated team of over 300 seasoned professionals.
What is the projected growth of the global drug safety monitoring market?
The global drug safety monitoring market is projected to reach USD 14.03 billion by 2032, with a compound annual growth rate (CAGR) of 8.3% from 2025 to 2032.
Why is drug safety important in public health?
Drug safety is crucial for safeguarding public health and safety, especially as oversight intensifies and the significance of monitoring drug safety escalates.
What qualifications do personnel need to meet regulatory requirements?
National competent authorities and the European Medicines Agency (EMA) mandate that personnel involved in drug safety must be qualified.
What role does the Eudravigilance database play in drug safety?
The Eudravigilance database disseminates drug safety information among competent authorities, emphasizing the importance of regulatory compliance.
What are the key compliance standards relevant to drug safety jobs?
Key compliance standards include Good Manufacturing Practices (GMP), International Organization for Standardization (ISO) standards, and Quality System Regulations (QSR).
What challenges does the pharmaceutical industry face regarding compliance?
The pharmaceutical industry faces significant compliance challenges, as highlighted by the FDA issuing thousands of observations for noncompliance.
How does AVS Life Sciences assist organizations with compliance?
AVS Life Sciences provides extensive consulting services to help organizations navigate the complexities of compliance and quality management in drug safety.
What is the significance of risk management in pharmaceuticals?
Risk management involves identifying, evaluating, and mitigating potential health risks associated with medicinal products, which is vital for patient safety throughout the drug lifecycle.
How has advanced analytics impacted risk management in pharmaceuticals?
The use of advanced analytics in risk management has led to significant reductions in negative event reporting durations and costs associated with preventable adverse events.
Can you provide an example of effective risk management?
A study across 42 hospitals reported a 42% reduction in medication errors, preventing approximately 1.2 million errors annually, demonstrating the effectiveness of structured risk management plans.
How does AVS Life Sciences support risk management strategies?
AVS Life Sciences provides comprehensive quality management and regulatory compliance solutions that help bolster risk management initiatives and improve patient well-being.
List of Sources
- AVS Life Sciences: Expertise in Drug Safety and Pharmacovigilance
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC3853665)
- coherentmarketinsights.com (https://coherentmarketinsights.com/market-insight/pharmacovigilance-market-1047)
- averyfairbank.com (https://averyfairbank.com/pharmacovigilance-what-is-it-and-why-is-it-important)
- 10 inspirational quotes for the pharma sector (https://pharmaceuticalmanufacturer.media/pharmaceutical-industry-insights/10-inspirational-quotes-for-the-pharma-sector)
- pharmacytimes.com (https://pharmacytimes.com/view/10-pharmacy-quotes-that-will-make-you-rethink-the-profession)
- Regulatory Compliance: Mastering Industry Standards and Guidelines
- 10 inspirational quotes for the pharma sector (https://pharmaceuticalmanufacturer.media/pharmaceutical-industry-insights/10-inspirational-quotes-for-the-pharma-sector)
- wilsonelser.com (https://wilsonelser.com/cybersecurity-data-privacy/news/captive-insurance-times-quotes-meer-at-the-2025-world-captive-forum)
- 7 Common Compliance Issues Faced By Pharmaceutical Industry (https://qualityze.com/blogs/7-common-compliance-issues-faced-by-pharmaceutical-industry)
- pharmtech.com (https://pharmtech.com/view/statistical-implications-cgmps-30-year-retrospective)
- Risk Management: Identifying and Mitigating Safety Concerns
- quotlr.com (https://quotlr.com/quotes-about-risk-management)
- numberanalytics.com (https://numberanalytics.com/blog/5-critical-stats-patient-safety-analytics-pharma-healthcare)
- atvigilx.com (https://atvigilx.com/benefit-risk-management-in-pharmacovigilance-for-better-drug-safety)
- managedhealthcareexecutive.com (https://managedhealthcareexecutive.com/view/risk-management-drug-safety-raise-challenges-sponsors-and-healthcare-providers)
- Quotes on Risk Management • Novel Investor (https://novelinvestor.com/quote-category/risk-management)
- Data Analysis: Interpreting Safety Data for Informed Decisions
- numberanalytics.com (https://numberanalytics.com/blog/5-critical-stats-patient-safety-analytics-pharma-healthcare)
- Top 20 Safety Quotes to Educate and Inspire Your Team (https://fluix.io/blog/safety-quotes)
- solutionsreview.com (https://solutionsreview.com/backup-disaster-recovery/data-privacy-week-2024-the-definitive-roundup-of-expert-quotes)
- 22 Workplace Safety Quotes From Leaders and Experts - AlertMedia (https://alertmedia.com/blog/workplace-safety-quotes)
- Biostatistics in Pharmacovigilance: Analyzing Safety Data (https://medipharmsolutions.com/blog/biostatistics-in-pharmacovigilance-analyzing-safety-data)
- Communication Skills: Collaborating Across Teams and Stakeholders
- booherresearch.com (https://booherresearch.com/dianna-booher-quotations)
- drugpatentwatch.com (https://drugpatentwatch.com/blog/effective-communication-strategies-for-generic-drug-teams?srsltid=AfmBOor39fzS82nyxvtsSAgmAkBqW_sDKxpOPeZPzPIT0F7E_hJt8wl0)
- 10 inspirational quotes for the pharma sector (https://pharmaceuticalmanufacturer.media/pharmaceutical-industry-insights/10-inspirational-quotes-for-the-pharma-sector)
- 20 Powerful Quotes on the Value of Clear Communication (https://medium.com/@tarunpatel/20-powerful-quotes-on-the-value-of-clear-communication-5133a23b042f)
- 50 Quotes on the Importance of Collaboration and Cooperation in Teamwork (https://inc.com/gordon-tredgold/50-quotes-on-the-importance-and-benefits-of-teamwork.html)
- Attention to Detail: Ensuring Accuracy in Safety Reporting
- Top 20 Safety Quotes To Improve Your Safety Culture (https://blog.safetyculture.com/industry-trends/top-20-safety-quotes-improve-safety-culture)
- Patient safety (https://who.int/news-room/fact-sheets/detail/patient-safety)
- Medication Errors | AMCP.org (https://amcp.org/concepts-managed-care-pharmacy/medication-errors)
- Contact Us | Outside US & Canada | Damotech (https://damotech.com/blog/inspirational-quotes-warehouse-safety)
- 22 Workplace Safety Quotes From Leaders and Experts - AlertMedia (https://alertmedia.com/blog/workplace-safety-quotes)
- Project Management: Overseeing Drug Safety Initiatives
- Project Management Statistics: 45 Stats You Can't Ignore (https://workamajig.com/blog/project-management-statistics)
- 55 Project Management Statistics of 2024 | Visual Planning (https://visual-planning.com/en/blog/project-management-statistics)
- Project Management Statistics and Trends for 2026 (https://plaky.com/learn/project-management/project-management-statistics)
- 139 Project Management Quotes to Inspire Your Next Project (https://plaky.com/blog/project-management-quotes)
- 16 Best Project Management Quotes (https://projectmanager.com/blog/10-best-project-management-quotes)
- Pharmacovigilance Systems: Monitoring Adverse Drug Reactions
- clinicaltherapeutics.com (https://clinicaltherapeutics.com/article/S0149-2918(18)30317-5/fulltext)
- regdesk.co (https://regdesk.co/drap-guidelines-on-national-pharmacovigilance-system-overview)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC3853665)
- journals.lww.com (https://journals.lww.com/cmre/fulltext/2022/12030/pharmacovigilance_study_of_spontaneous_adverse.3.aspx)
- ema.europa.eu (https://ema.europa.eu/en/human-regulatory-overview/post-authorisation/pharmacovigilance-post-authorisation/pharmacovigilance-system-questions-answers)
- Ethical Considerations: Upholding Integrity in Drug Safety
- hcplive.com (https://hcplive.com/view/medical-ethics-unpacked-glp-1-allocation-and-challenges-of-equitable-access)
- techtarget.com (https://techtarget.com/pharmalifesciences/feature/Dissecting-Ethical-Principles-in-the-Pharmaceutical-Industry)
- pharmaoffer.com (https://pharmaoffer.com/blog/the-top-5-ethical-controversies-in-the-pharmaceutical-industry)
- Ethics Quotes and Morality Quotes Related to Decision Making (https://decision-making-solutions.com/ethics_quotes.html)
- Continuous Learning: Adapting to Evolving Industry Standards
- 10 inspirational quotes for the pharma sector (https://pharmaceuticalmanufacturer.media/pharmaceutical-industry-insights/10-inspirational-quotes-for-the-pharma-sector)
- 7 Statistics That Highlight the Value of Continuous Learning (https://edume.com/blog/continuous-learning-in-the-workplace)
- 7 Continuous Learning Quotes, That'll Inspire You in 2026! | Vouch (https://vouchfor.com/blog/continuous-learning-quotes)
- 90+ Employee Training Statistics in 2025: Investing in Employee Development | Shortlister (https://myshortlister.com/insights/employee-training-statistics)
- 18 Powerful Quotes To Inspire You To Embrace Continuous Learning (https://linkedin.com/pulse/18-powerful-quotes-inspire-you-embrace-continuous-learning-jenkins)
