7 Ways How AVS Handles Facility Registration and FDA Updates for Clients

Introduction
AVS Life Sciences stands at the forefront of regulatory compliance, expertly navigating the complexities of facility registration and FDA updates. In an era where the industry faces increasing scrutiny and evolving regulations, organizations are compelled not only to meet compliance standards but also to excel in operational efficiency. This article explores the innovative strategies employed by AVS, showcasing how their tailored solutions and cutting-edge technology empower clients to surmount regulatory challenges.
What key practices can transform compliance management into a streamlined process?
How can organizations leverage these insights to enhance their own regulatory frameworks?
The answers lie within the expertise of AVS Life Sciences.
AVS Life Sciences: Streamlining Facility Registration Processes
AVS Life Sciences demonstrates how AVS handles and by addressing the compliance challenges faced by facilities in the registration process with a systematic approach. By leveraging extensive knowledge of , including the and the , our team of specialists—comprising over 300 seasoned associates worldwide—assists customers in preparing and submitting essential documentation. This guarantees adherence to FDA requirements, particularly in how AVS handles and , with the impending enforcement for product listing starting on July 1, 2024.
Through , AVS experts guide individuals in understanding how AVS handles and , helping them avoid common pitfalls. Our customized strategies not only expedite the registration process but also enhance the overall regulatory posture of the facilities involved. By applying best practices, such as and emphasizing in , while adhering to , AVS improves efficiency in maneuvering through the oversight environment. This ultimately supports customers in achieving their compliance objectives.
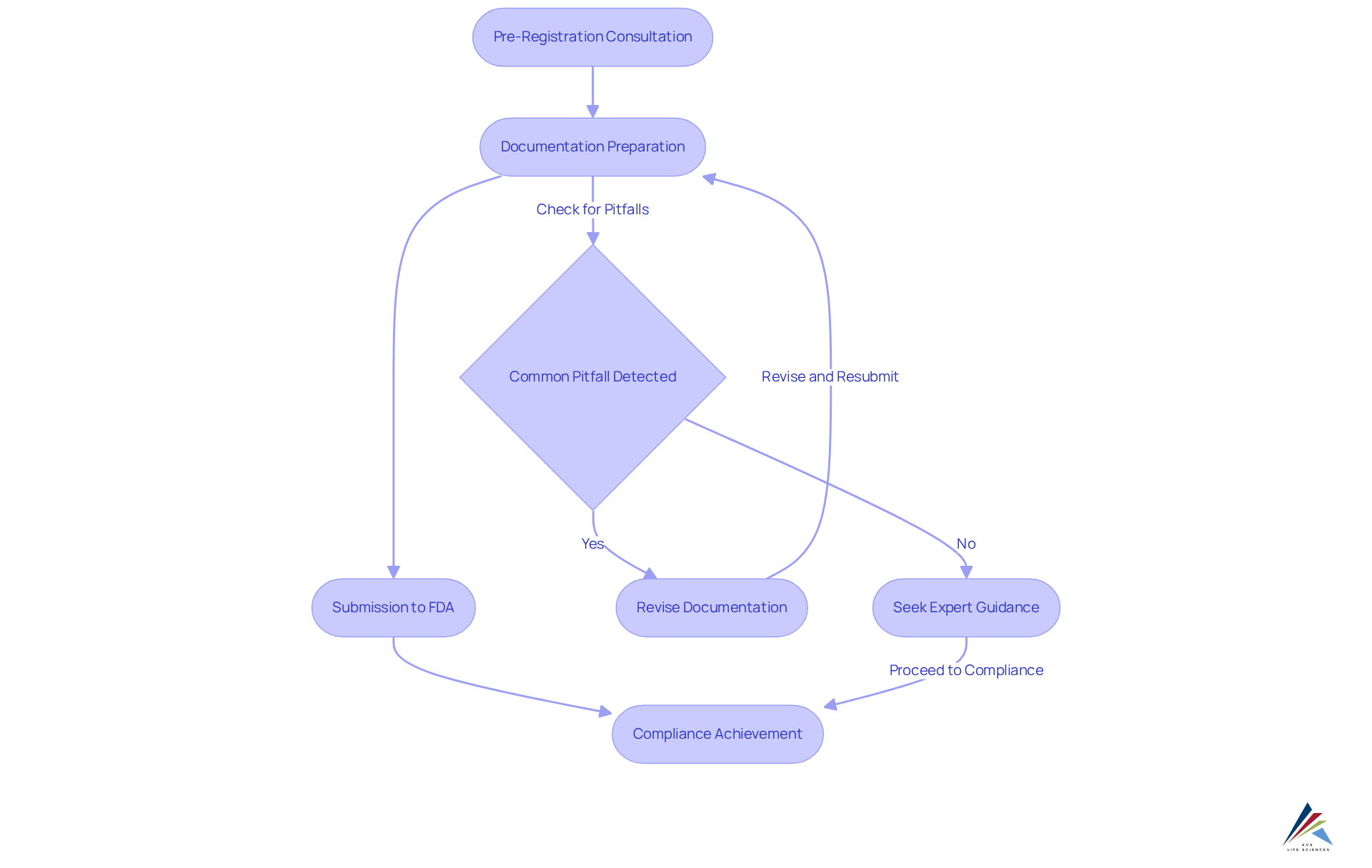
Understanding FDA Regulations for Facility Registration
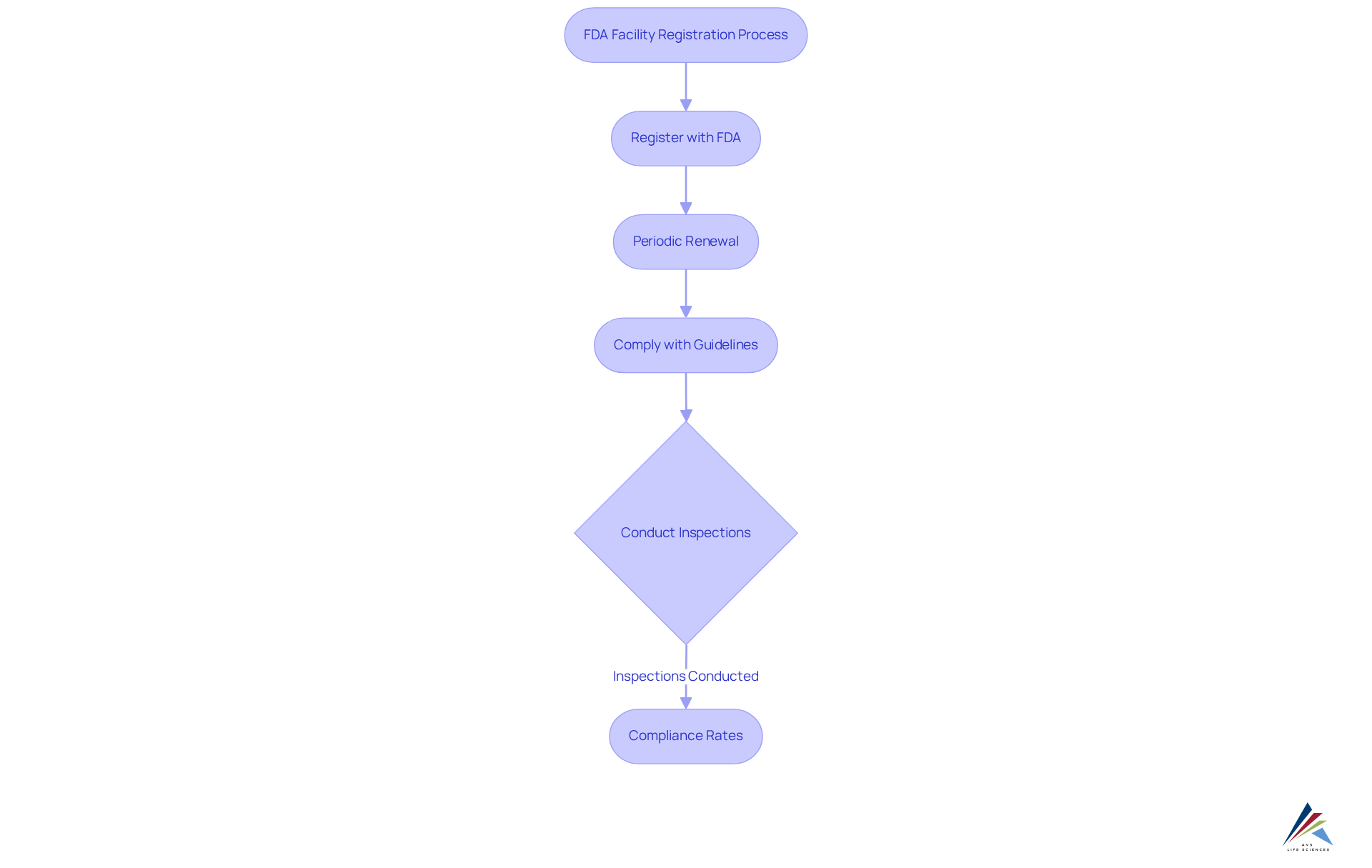
The FDA mandates that all facilities engaged in the manufacturing, processing, packing, or holding of food, drugs, and medical devices register with the agency. This registration is not merely a one-time obligation; it requires periodic renewal, and facilities must comply with specific guidelines outlined in the Federal Food, Drug, and Cosmetic Act. Notably, compliance rates for FDA s have shown remarkable improvement, with the FDA conducting 18,169 inspections in 2023—a 17.6% increase from the previous year.
AVS Life Sciences provides customers with comprehensive insights into how AVS handles and for clients, highlighting the importance of understanding their responsibilities to maintain and avoid . A transformative case study exemplifies AVS's successful enhancement of a biotechnology . Through our expertise in and , the customer progressed from a to a Level 2 , significantly boosting their manufacturing capabilities while strictly following guidelines. As one compliance officer aptly stated, ' is not just about compliance; it's about ensuring the safety and efficacy of our products.'
This perspective highlights the vital role that AVS plays in how AVS handles and for clients, helping individuals navigate the complexities of effectively and illustrating the substantial ramifications of non-adherence.
Leveraging Technology for Efficient FDA Updates
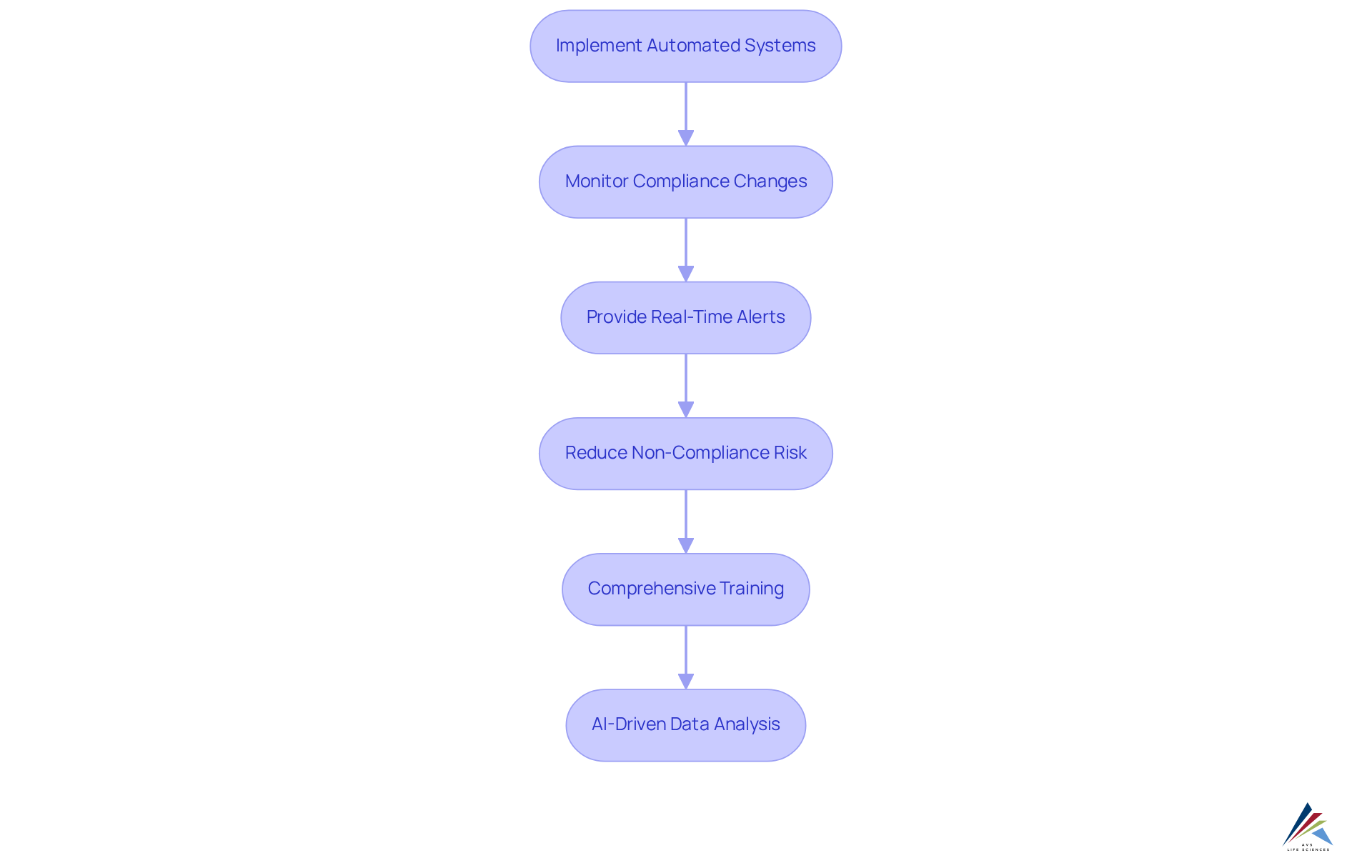
AVS Life Sciences utilizes cutting-edge technology to enhance how AVS handles and for clients, effectively addressing the pressing faced by the industry. By implementing that monitor compliance changes, clients benefit from . This proactive strategy significantly reduces the , ensuring that facilities consistently adhere to the latest regulations. In fact, AI-driven real-time monitoring can decrease adherence issues by 40%, demonstrating the effectiveness of AVS's approach.
Furthermore, AVS provides on these technologies, emphasizing the importance of mastering prior to FDA inspections, as recommended by UL Solutions. Industry leaders stress that automation plays a vital role in enhancing compliance, with . For instance, over 60% of top pharma companies have adopted AI-based automation for at least one stage of drug manufacturing, showcasing the industry's shift towards technology.
Additionally, by as much as 35%, further illustrating the impact of automation. AVS Life Sciences exemplifies this method, showcasing how technology can transform compliance management into a more efficient and trustworthy process. By engaging with AVS, organizations can learn how AVS handles and for clients, enabling them to navigate the complexities of compliance and elevate their operational standards to meet regulatory expectations.
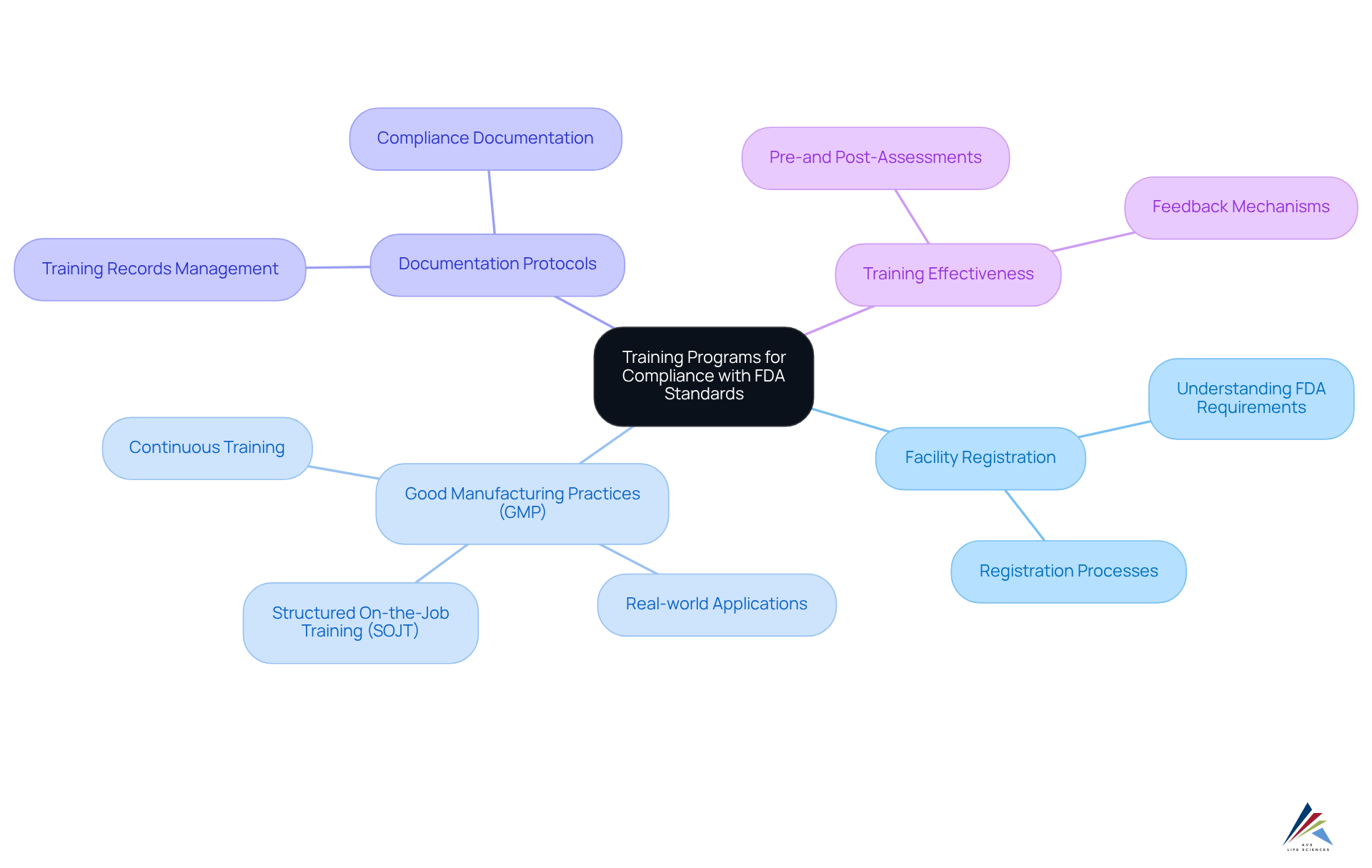
Training Programs for Compliance with FDA Standards
AVS Life Sciences presents comprehensive training programs meticulously crafted to enlighten clients about how AVS handles for clients, along with . These programs cover , including how AVS handles for clients, , and .
By equipping employees with essential knowledge and skills, AVS cultivates a culture of adherence within organizations, significantly mitigating the risk of violations and enhancing operational efficiency. Current trends underscore that , integrating real-world applications and recent industry developments to sustain employee interest and knowledge retention.
Insights from regulatory trainers highlight the necessity of and regular assessments to ensure that employees not only comprehend but can also implement FDA standards proficiently. This thorough approach to training fulfills legal standards while preparing organizations for in navigating the complexities of how AVS handles for clients.
A : AVS assisted a leading biotechnology company in upgrading its GMP facility, confronting challenges such as initial test result anomalies due to improperly installed barcode scanners. By addressing these issues and capturing vital lessons learned, AVS ensured that the staff was well-trained in . This partnership not only facilitated adherence but also allowed the client to focus on developing life-saving medications, underscoring the direct impact of effective training on operational success.
Effective Documentation Management for Regulatory Compliance
Efficient is paramount for navigating regulatory landscapes, and how AVS handles and for clients is what keeps AVS Life Sciences at the forefront of this critical area. The company employs that guarantee all , readily accessible, and consistently updated. This encompasses:
- The development of standardized templates for essential documents
- Regular audits of documentation practices
- Extensive training for staff on proper documentation protocols
that efficient documentation not only streamlines the audit process but also significantly , ultimately yielding improved outcomes in inspections. AVS ensures its documentation practices meet the highest criteria by adhering to FDA regulations and , which illustrates how AVS handles and for clients. For instance, by 82%, exemplifying the tangible benefits of a robust system. By prioritizing these practices, AVS demonstrates how AVS handles and for clients, empowering them to mitigate risks associated with non-compliance and enhancing their preparedness for successful audits.
Navigating Challenges in Regulatory Compliance
Organizations frequently encounter a myriad of challenges in , particularly within the life sciences sector, where regulations evolve rapidly and resource constraints are common. A significant 70% of corporate risk and regulatory experts have noted a transition toward a more , underscoring the imperative for organizations to adapt effectively.
AVS Life Sciences equips clients to navigate these complexities by providing tailored solutions, specifically focusing on how AVS handles and for clients to meet their specific needs. This process includes:
- Conducting thorough gap assessments to pinpoint deficiencies in
- Offering strategic insights on
- Delivering continuous support to ensure adherence
As one consultant aptly stated, 'Overcoming legal obstacles demands not only comprehending the guidelines but also foreseeing changes and preparing accordingly.'
AVS's commitment to proactive illustrates how AVS handles and for clients, empowering them to thrive amidst while ensuring alignment with their core business objectives.
Preparing for Audits and Inspections: Best Practices
Thorough preparation for audits and inspections is essential in the life sciences field, where adherence to is vital. AVS Life Sciences advocates for several best practices to bolster .
- Conducting stands out as a key strategy; this approach allows organizations to identify potential issues before the actual audit occurs. Such a proactive strategy not only emphasizes areas requiring enhancement but also fosters employee confidence in addressing .
- Maintaining is crucial, as it facilitates a smoother audit process.
- Ensuring that all staff are well-trained and aware of their specific roles during an audit can significantly enhance overall readiness.
- Furthermore, is another critical component; fostering transparency can lead to a more collaborative and constructive audit experience.
Statistics reveal that organizations employing report a marked improvement in . Notably, 89% of professionals acknowledge that such practices expedite time-to-compliance across multiple frameworks. By implementing these best practices, organizations can effectively enhance their and minimize the risk of non-compliance findings, ultimately safeguarding their reputation and operational integrity.
Enhancing Communication for Successful Facility Registration
is paramount for clients to understand how AVS handles . AVS Life Sciences emphasizes how AVS handles for clients by ensuring between customers and governing entities, keeping all parties informed and coordinated throughout the registration process. This commitment encompasses:
- Regular updates on application statuses
- Prompt responses to inquiries
By fostering a culture of transparency and responsiveness, AVS empowers clients to navigate the complexities of how AVS handles for clients, resulting in .
Professionals in affairs stress that not only improves adherence but also fosters trust, which is vital for successful interactions with oversight agencies. For instance, AVS Life Sciences has successfully facilitated numerous client-regulatory body communications, illustrating how AVS handles for clients, which demonstrates that proactive engagement can lead to smoother approvals and a more streamlined registration process.
A transformative case study illustrates this: AVS assisted a leading biotechnology company in upgrading their manufacturing space from a Biosafety Level 1 GMP facility to a Level 2 GMP facility. Throughout this project, AVS upheld , which was vital for tackling challenges and ensuring adherence to standards.
AVS utilized specific methodologies such as gap analysis and customized communication strategies to ensure all compliance requirements were met effectively. Through these efforts, AVS Life Sciences illustrates how can greatly influence success in meeting regulations.
Tailored Solutions for Unique Client Needs in Facility Registration
AVS Life Sciences understands that how AVS handles and FDA updates for clients is not a one-size-fits-all process. Each customer presents unique challenges and requirements, prompting the company to offer that address specific organizational needs. This includes:
- designed to enhance
- Specialized documentation templates that streamline the registration process
- Dedicated support throughout every stage of registration
With expertise in GXP, , and the development of , AVS ensures that customers are well-equipped to meet . Moreover, AVS emphasizes effective documentation methods and addresses to further bolster adherence. For instance, AVS has developed training modules that highlight the intricacies of , significantly improving success rates among customers.
Industry experts assert, 'Tailored regulatory strategies are essential for navigating the complexities of effectively.' By prioritizing customized services and leveraging their extensive , AVS empowers customers to understand how AVS handles and FDA updates for clients, allowing them to navigate the complexities with confidence and ensuring they meet efficiently.
Continuous Improvement in Regulatory Compliance Practices
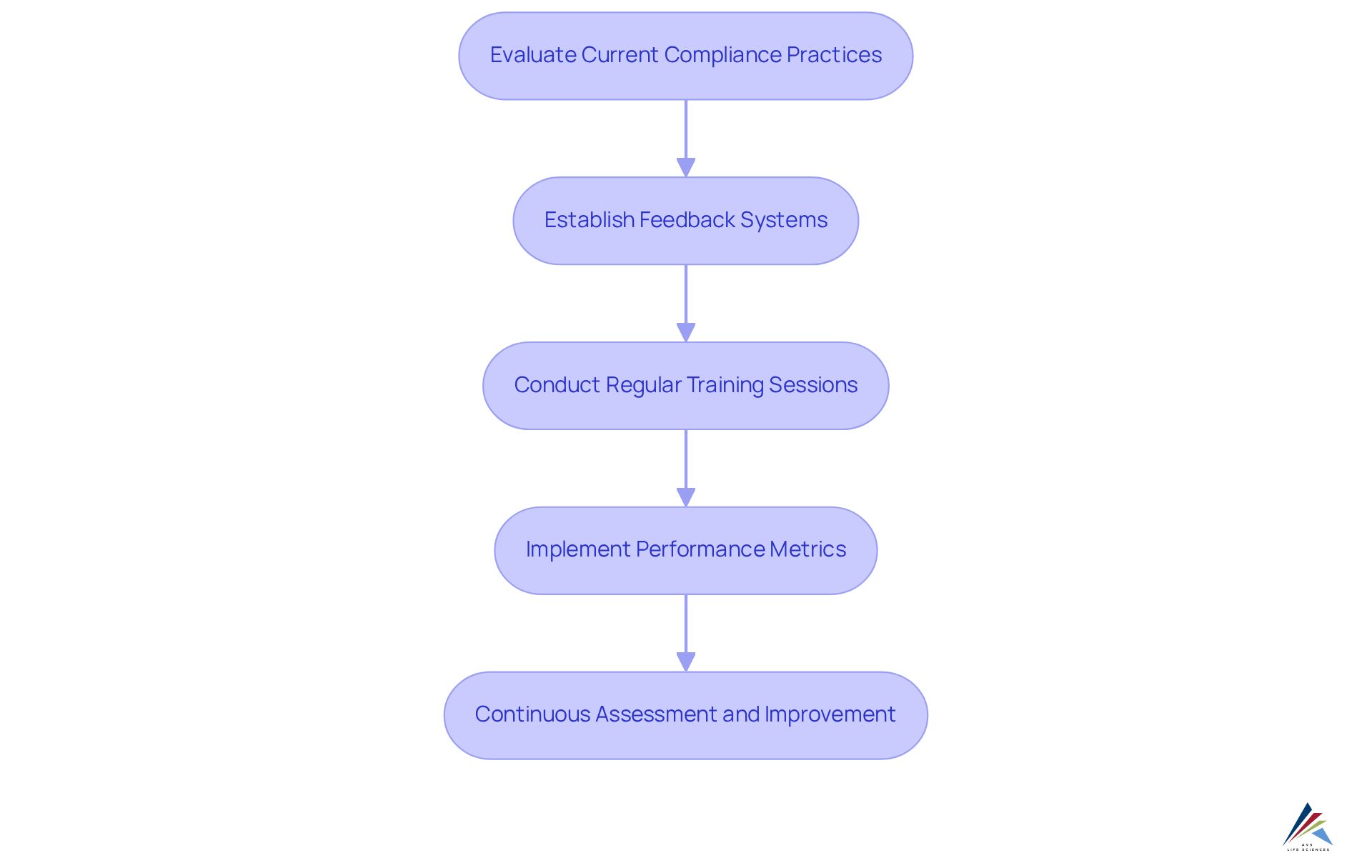
At AVS Life Sciences, continuous improvement transcends mere principle; it stands as a fundamental operational strategy. The company actively empowers clients to routinely evaluate and enhance their adherence practices, ensuring alignment with evolving regulations and industry standards. This proactive approach includes the establishment of robust , essential for assessing adherence effectiveness. Regular play a pivotal role, equipping staff with the necessary knowledge to navigate regulatory challenges effectively. Performance metrics are employed to measure the success of these initiatives, enabling organizations to pinpoint areas for enhancement.
By fostering a culture of perpetual improvement, AVS enables organizations not only to meet current regulatory standards but also to anticipate and prepare for future adherence challenges. This commitment to adaptability is vital in a landscape where 60% of executives report increased investment in , underscoring its growing importance. Organizations that implement effective , as evidenced by numerous case studies, have experienced significant improvements in adherence outcomes, highlighting the tangible benefits of AVS's methodology. As industry leaders emphasize the necessity of , AVS Life Sciences positions itself as a dedicated partner by showcasing how AVS handles and for clients, assisting them in navigating the complexities of . To fully leverage the advantages of AVS's services, officers must engage in regular assessments and training, ensuring their organizations remain at the forefront of .
Conclusion
AVS Life Sciences exemplifies a comprehensive approach to managing facility registration and FDA updates, tailored to meet the unique needs of each client. By integrating regulatory expertise with advanced technology, AVS not only simplifies the registration process but also ensures that clients remain compliant with evolving FDA standards. This commitment to personalized service and strategic guidance positions AVS as a vital partner in navigating the complexities of regulatory compliance.
Throughout the article, key strategies such as:
- Pre-registration consultations
- Effective documentation management
- Continuous training programs
have been highlighted. AVS's use of technology, including automated compliance monitoring and AI-driven data analysis, significantly enhances operational efficiency and reduces the risk of non-compliance. Moreover, the emphasis on open communication fosters a collaborative environment that is essential for successful interactions with regulatory bodies.
In an industry where regulatory landscapes are constantly shifting, the importance of proactive compliance management cannot be overstated. Organizations are encouraged to leverage AVS Life Sciences' expertise to not only meet current standards but also to anticipate future challenges. By prioritizing continuous improvement and tailored solutions, clients can enhance their operational integrity and ensure that they are well-prepared for upcoming FDA requirements. Engaging with AVS is not just a step toward compliance; it is a strategic investment in the future success and sustainability of any facility within the life sciences sector.
Frequently Asked Questions
What is the role of AVS Life Sciences in facility registration and FDA updates?
AVS Life Sciences assists clients in navigating the facility registration process and ensuring compliance with FDA requirements by providing systematic approaches and leveraging extensive knowledge of regulatory frameworks.
What regulatory frameworks does AVS Life Sciences focus on?
AVS Life Sciences focuses on regulatory frameworks such as the Food Safety Modernization Act (FSMA) and the Bioterrorism Act to help clients with facility registration and compliance.
How does AVS Life Sciences help clients avoid common pitfalls in the registration process?
AVS Life Sciences offers pre-registration consultations where experts guide clients in understanding the registration process, helping them avoid common pitfalls and expedite their registration.
What strategies does AVS Life Sciences employ to enhance regulatory compliance?
AVS Life Sciences employs customized strategies, best practices like Standard Operating Procedures (SOPs), and emphasizes data integrity in Computer System Validation (CSV) to enhance regulatory compliance.
What are the FDA's requirements for facility registration?
The FDA requires all facilities involved in the manufacturing, processing, packing, or holding of food, drugs, and medical devices to register with the agency, which includes periodic renewals and adherence to specific guidelines.
How has the compliance rate for FDA facility registrations changed recently?
Compliance rates for FDA facility registrations have improved, with the FDA conducting 18,169 inspections in 2023, marking a 17.6% increase from the previous year.
Can you provide an example of AVS Life Sciences' impact on a client's facility?
AVS Life Sciences successfully enhanced a biotechnology GMP facility's capabilities, progressing it from a Biosafety Level 1 to a Level 2 GMP facility while ensuring compliance with quality assurance standards.
How does AVS Life Sciences use technology in the registration process?
AVS Life Sciences utilizes cutting-edge technology, including automated systems that monitor compliance changes and provide real-time alerts, significantly reducing the risk of non-compliance.
What are the benefits of AI-driven real-time monitoring in compliance management?
AI-driven real-time monitoring can decrease adherence issues by 40% and reduce submission errors by as much as 35%, demonstrating its effectiveness in enhancing compliance management.
What training does AVS Life Sciences provide regarding compliance technologies?
AVS Life Sciences offers comprehensive training on compliance technologies, emphasizing the importance of mastering essential competencies prior to FDA inspections.
List of Sources
- AVS Life Sciences: Streamlining Facility Registration Processes
- azquotes.com (https://azquotes.com/quotes/topics/regulation.html)
- fda.gov (https://fda.gov/food/guidance-regulation-food-and-dietary-supplements/registration-food-facilities-and-other-submissions)
- avslifesciences.com (https://avslifesciences.com/blog-post/7-key-insights-on-csv-full-form-in-pharma-compliance)
- cosmetic.chemlinked.com (https://cosmetic.chemlinked.com/news/cosmetic-news/us-fda-unveils-latest-data-on-facility-registration-and-product-listing-under-mocra)
- Understanding FDA Regulations for Facility Registration
- foley.com (https://foley.com/insights/publications/2024/08/2024-cpsc-fda-enforcement-trends)
- Remarks, FDLI Food Enforcement and Compliance Conference (https://fda.gov/news-events/speeches-fda-officials/remarks-food-and-drug-law-institutes-food-enforcement-and-compliance-conference-04122018)
- prnewswire.com (https://prnewswire.com/news-releases/fda-removed-28-of-food-facility-registrations-from-its-database-in-2017-registrar-corp-reports-300440755.html)
- brewersassociation.org (https://brewersassociation.org/association-news/unique-facility-identifier-now-required-for-fda-facility-registration-and-renewals)
- Leveraging Technology for Efficient FDA Updates
- patentpc.com (https://patentpc.com/blog/ai-in-pharma-manufacturing-how-automation-is-changing-drug-production-market-stats)
- 37 Quotes on Health Care and Health Tech from 2016 (https://medium.com/@r_scott_munro/37-quotes-on-health-care-and-health-tech-from-2016-e34576edfc1f)
- ul.com (https://ul.com/resources/fda-key-points-regulated-life-science-industries)
- scigeniq.com (https://scigeniq.com/always-audit-ready-how-automated-systems-transform-pharma-compliance)
- xybion.com (https://xybion.com/blog/strategies-to-ensure-your-qms-meets-fda-and-iso-compliance)
- Training Programs for Compliance with FDA Standards
- complianceonline.com (https://complianceonline.com/resources/fda-compliance-regulatory-training.html)
- biopharminternational.com (https://biopharminternational.com/view/if-training-so-easy-why-isnt-everyone-compliance)
- Effective Training Metrics for Quality and Performance (https://compliancequest.com/blog/driving-organizational-efficiency-and-performance-through-effective-training-metrics)
- gmptrends.com (https://gmptrends.com/the-importance-of-employee-training)
- fdalisting.com (https://fdalisting.com/customized-training)
- Effective Documentation Management for Regulatory Compliance
- 100+ Compliance Statistics You Should Know in 2025 (https://sprinto.com/blog/compliance-statistics)
- numberanalytics.com (https://numberanalytics.com/blog/stats-regulatory-analytics-boost-healthcare-safety)
- 100 Document Management Statistics to Make You Rethink Your Processes in 2025 (https://filecenter.com/blog/document-management-statistics)
- 115 Compliance Statistics You Need To Know in 2023 - Drata (https://drata.com/blog/compliance-statistics)
- biotech.com (https://biotech.com/2023/02/02/maintaining-project-documentation-and-records-in-life-science)
- Navigating Challenges in Regulatory Compliance
- 25 Critical Stats Every Chief Compliance Officer Needs to Know in 2025 (https://complianceandrisks.com/blog/25-critical-stats-every-chief-compliance-officer-needs-to-know-in-2025)
- 7 Compliance Statistics and What They Mean For You - Thoropass (https://thoropass.com/blog/compliance/7-compliance-statistics-and-what-they-mean-for-you)
- 115 Compliance Statistics You Need To Know in 2023 - Drata (https://drata.com/blog/compliance-statistics)
- 130+ Compliance Statistics & Trends to Know for 2026 (https://secureframe.com/blog/compliance-statistics)
- Key Compliance Statistics & Insights For 2025 | Zluri (https://zluri.com/blog/key-compliance-statistics-and-insights-for-2024)
- Preparing for Audits and Inspections: Best Practices
- 130+ Compliance Statistics & Trends to Know for 2026 (https://secureframe.com/blog/compliance-statistics)
- 171 Motivational Quotes for Accountants and CPAs [2026] (https://acecloudhosting.com/blog/motivational-quotes-accountants)
- 10 Metrics to Measure Compliance Training (https://trainingindustry.com/articles/compliance/10-metrics-to-measure-compliance-training)
- 115 Compliance Statistics You Need To Know in 2023 - Drata (https://drata.com/blog/compliance-statistics)
- Audit Quotes (8 quotes) (https://goodreads.com/quotes/tag/audit)
- Enhancing Communication for Successful Facility Registration
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC10442955)
- nationalfacilitiesdirect.com (https://nationalfacilitiesdirect.com/how-to-enhance-communication-in-facilities-management)
- Communications Statistics You Need to Know in 2026 (https://sociabble.com/blog/employee-communications/communications-statistics)
- brosix.com (https://brosix.com/blog/workplace-communication-statistics)
- expertmarket.com (https://expertmarket.com/phone-systems/workplace-communication-statistics)
- Tailored Solutions for Unique Client Needs in Facility Registration
- toolsense.io (https://toolsense.io/studies-reports/facility-management-statistics)
- fortunebusinessinsights.com (https://fortunebusinessinsights.com/industry-reports/facility-management-market-101658)
- rfm-group.com (https://rfm-group.com/50-reasons-to-use-a-facilities-management-company-a-new-fms-guide)
- Facilities Management Stats: Market Size & Future | ProfileTree (https://profiletree.com/facilities-management-stats-market-size-future)
- qualtrics.com (https://qualtrics.com/blog/research-quotes)
- Continuous Improvement in Regulatory Compliance Practices
- 100+ Compliance Statistics You Should Know in 2025 (https://sprinto.com/blog/compliance-statistics)
- 115 Compliance Statistics You Need To Know in 2023 - Drata (https://drata.com/blog/compliance-statistics)
- numberanalytics.com (https://numberanalytics.com/blog/stats-regulatory-analytics-boost-healthcare-safety)
- Key Compliance Statistics & Insights For 2025 | Zluri (https://zluri.com/blog/key-compliance-statistics-and-insights-for-2024)
- 130+ Compliance Statistics & Trends to Know for 2026 (https://secureframe.com/blog/compliance-statistics)
