10 Key Insights for Navigating the CGMP Environment

Introduction
The landscape of pharmaceutical manufacturing is becoming increasingly complex, with compliance with Current Good Manufacturing Practices (CGMP) more critical than ever. As regulatory scrutiny intensifies, organizations must not only grasp these standards but also skillfully navigate the challenges they present. This article explores ten key insights that illuminate the path to effective CGMP adherence, providing invaluable guidance for industry professionals aiming to enhance product safety and operational efficiency. How can organizations leverage these insights to not only meet regulatory demands but also secure a competitive edge in an evolving market?
AVS Life Sciences: Key Insights on Current Good Manufacturing Practices (CGMP)
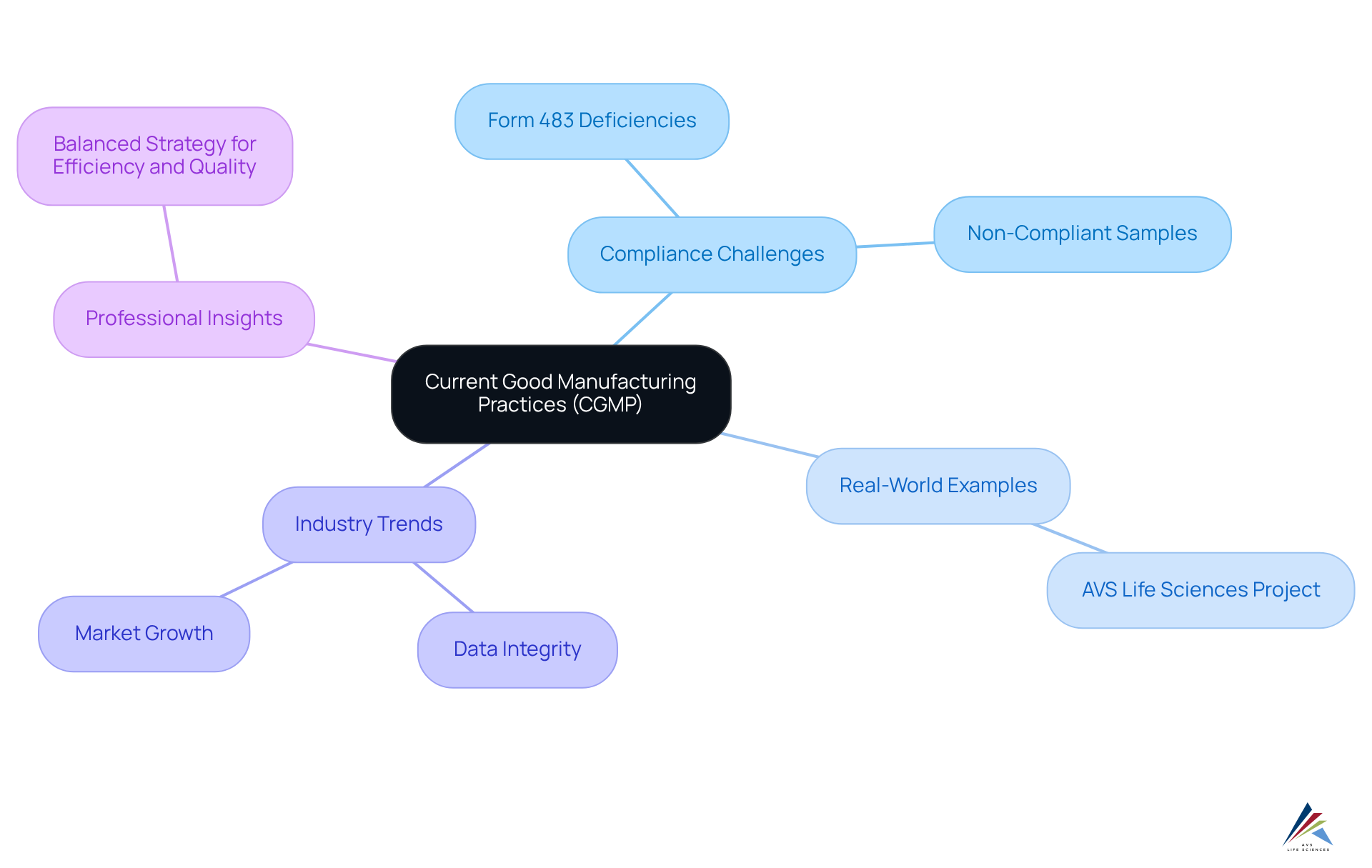
AVS Life Sciences provides in a , empowering clients in the pharmaceutical and biotechnology sectors to navigate effectively. The significance of is underscored by the FDA's documentation of deficiencies in over 5,000 Form 483s, indicating widespread compliance challenges. In fiscal year 2021, the percentage of non-compliant samples escalated to 35%, a stark increase from 16% the prior year, underscoring the .
Real-world examples demonstrate the successful application of . For instance, in upgrading its manufacturing area to align with , completing the project on time and within budget. This case exemplifies how adherence to the not only mitigates but also enhances operational efficiency and product integrity.
Current trends indicate a growing emphasis on , particularly as the biologics market is projected to expand at a compound annual growth rate of 15% by 2025. The FDA's New Inspection Protocol Project (NIPP) aims to improve inspection efficacy through a targeted, data-driven approach, reflecting the industry's shift towards more sophisticated regulatory practices.
Professional insights highlight the necessity of a , with industry leaders advocating for a balanced strategy that emphasizes both efficiency and quality enhancement. Organizations that commit to not only safeguard public health but also bolster their market reputation, ultimately leading to greater success in a competitive landscape.
Understanding CGMP: Definition and Core Principles
Current (CGMP) are regulations enforced by the FDA to ensure that manufacturing processes and facilities are designed, monitored, and controlled effectively. The core principles of CGMP encompass several critical aspects:
- Clean and Hygienic Environments: Maintaining a clean manufacturing environment is essential to prevent contamination and ensure product safety. Businesses that adhere to implement strict sanitation protocols and consistently educate staff on hygiene methods.
- Consistent Production and Quality Control: mandate that products are consistently produced and controlled according to established quality standards. This includes conducting in-process controls and testing to verify that each batch meets predefined specifications.
- Robust Documentation Practices: is a cornerstone of . It provides an audit trail for regulatory verification, ensuring that all processes—from raw material sourcing to final product release—are well-documented and traceable.
A significant example of is , where they assisted a pharmaceutical manufacturer in enhancing their facility from a Biosafety Level 1 to a Level 2 GMP environment for the production of lentivirus vector material. This transformation involved a , equipment installation, and , ensuring and GXP standards. AVS Life Sciences' meticulous documentation practices throughout this project demonstrated full traceability, which was positively received by the client's quality assurance team.
Statistics indicate that failure to adhere to can result in major consequences, such as costly product recalls and FDA-issued warning letters. For instance, the FDA has not recognized any process models that validate foundational assumptions throughout the manufacturing process, underscoring the necessity for .
Examples of companies successfully maintaining under the CGMP environment include those that have adopted advanced manufacturing technologies and continuous improvement practices. These organizations not only comply with regulatory standards but also enhance operational efficiency and foster consumer trust through their dedication to excellence and safety.

Importance of CGMP: Ensuring Product Safety and Quality
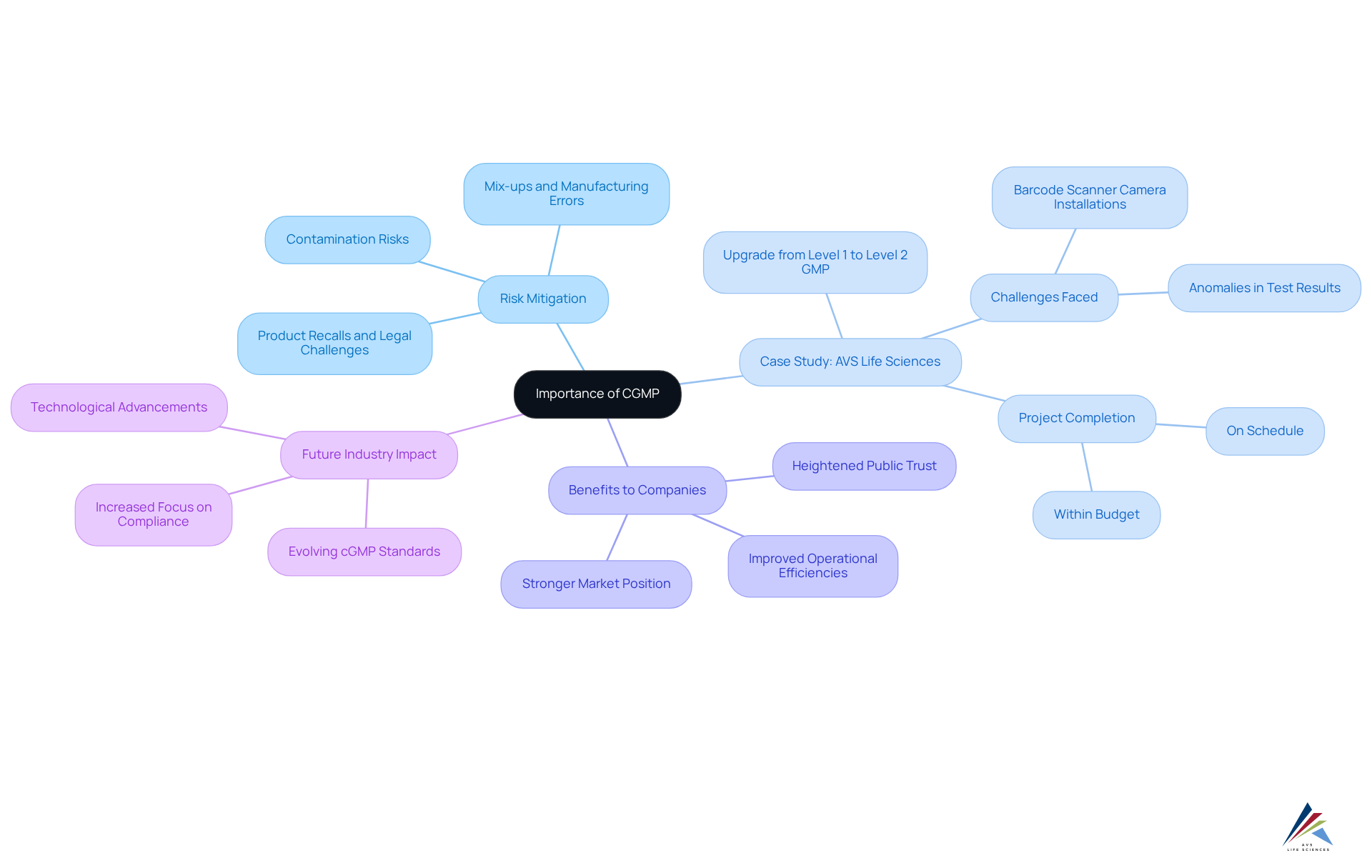
The is vital for ensuring that pharmaceutical products are not only safe and effective but also meet the . By rigorously adhering to a , organizations can significantly mitigate the . This commitment not only protects consumer health but also enhances the credibility and reputation of the company in the competitive pharmaceutical landscape.
A transformative case study exemplifying this is ' successful upgrade of a biotechnology client's from a Biosafety Level 1 GMP to a Level 2 GMP. This project encountered challenges, such as the oversight of barcode scanner camera installations, which initially resulted in anomalies in test results. However, the project was completed on schedule and within budget, demonstrating how effective assurance practices can bolster compliance and operational efficiency.
Companies that have embraced a , such as , report improved and heightened public trust, which contribute to a stronger market position. As the industry evolves in 2025, the influence of cGMP on product quality will become increasingly evident, with specific statistics indicating that organizations adhering to these practices experience fewer product recalls and legal challenges. Ultimately, the transcends mere regulatory compliance; it represents a strategic advantage that fosters and fortifies the integrity of the pharmaceutical supply chain.
FDA Compliance Assessment: How the FDA Evaluates CGMP Adherence
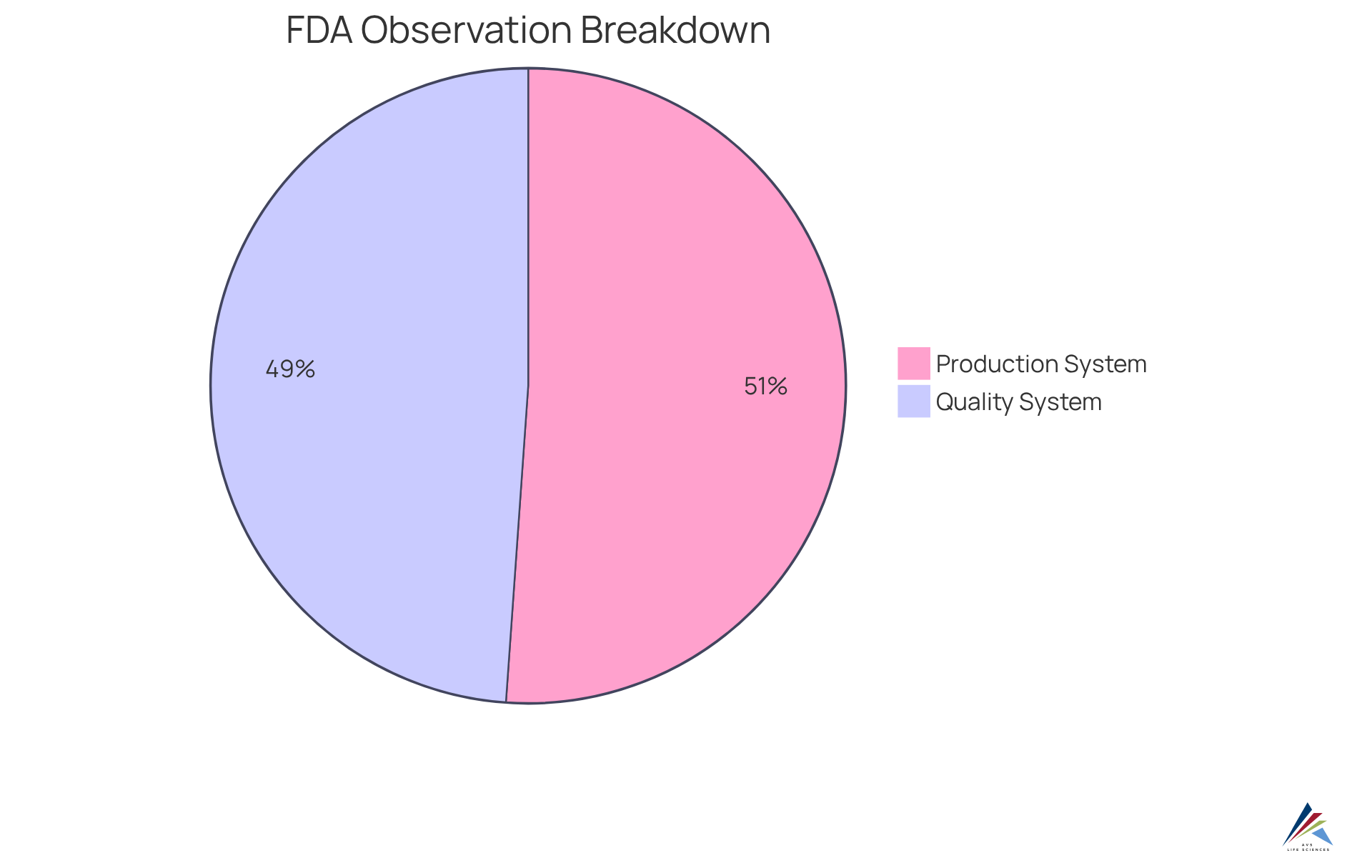
The FDA evaluates adherence to the through of manufacturing facilities, focusing on documentation, processes, and conformity to established regulations. Organizations must be prepared to demonstrate their within a , which involves maintaining detailed records that facilitate a smooth inspection process. Notably, statistics reveal that:
- The Production System accounted for 27.4% of total from 2018 to 2021.
- The closely followed with 26.2%.
This highlights during audits. Insights from regulatory specialists suggest that a proactive strategy for in a can significantly enhance the likelihood of successfully passing inspections. Effective audits often depend on an organization’s ability to showcase in a and . This can lead to favorable evaluations from FDA inspectors and a reduction in identified deficiencies.
Consequences of Non-Compliance: Risks to Drug Safety
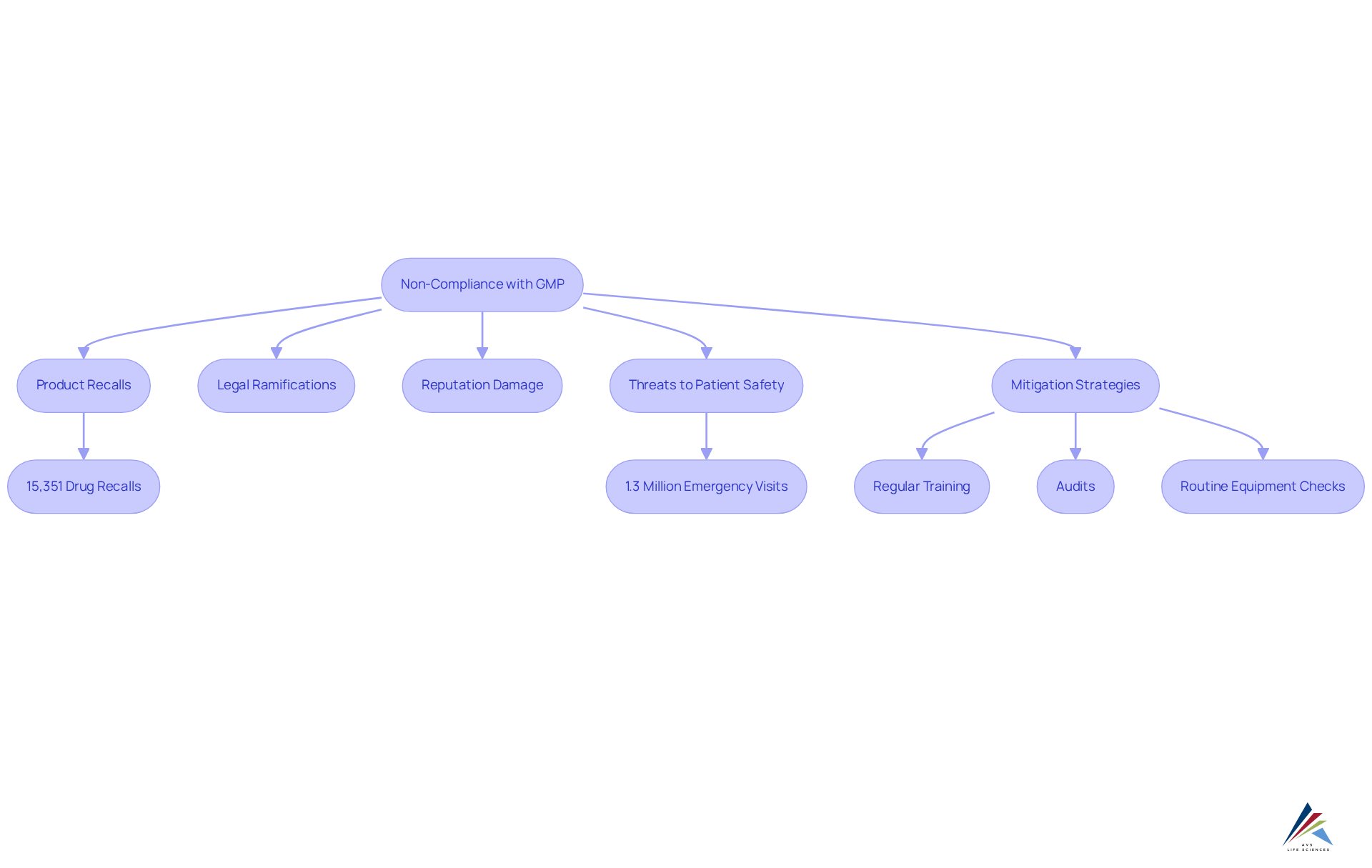
Non-compliance with in a poses severe consequences, including product recalls, legal ramifications, and detrimental impacts on a company's reputation. The FDA has reported over 15,351 in the U.S. since 2012, with 94% occurring nationally, underscoring the prevalence of . Such violations compromise the integrity of pharmaceutical products and significantly threaten patient safety, potentially leading to adverse health outcomes.
For example, result in approximately 1.3 million emergency department visits annually, highlighting the critical need for stringent adherence to the . Organizations must recognize that maintaining these standards in a transcends mere regulatory obligation; it is a moral imperative to protect public health and ensure product safety.
The fallout from violations within a extends beyond immediate legal and financial repercussions, fostering long-term trust deficits with consumers and stakeholders, ultimately jeopardizing business viability. A transformative case study from exemplifies this: by assisting a leading biotechnology company in upgrading their manufacturing area from a Biosafety Level 1 GMP facility to a Level 2 GMP facility, AVS enabled the client to focus on developing life-saving medicines while upholding rigorous .
During this transition, discrepancies were identified in test results due to improperly installed barcode scanner cameras, which had been initially overlooked. This experience prompted the QC laboratory and Quality teams to reassess their processes, resulting in enhanced testing protocols and improved team communication.
To mitigate risks, regulatory officers should implement regular training and audits, drawing lessons from like that of . A practical recommendation for regulatory officers is to establish routine examinations of equipment and procedures to identify potential issues before they escalate into .
FDA Actions Against CGMP Violations: Protecting Public Health
The FDA wields significant authority in addressing violations within the , employing a range of measures such as issuing warning letters, imposing penalties, and even shutting down non-compliant production facilities. In FY23 alone, the agency issued 180 , indicating a marked increase in enforcement efforts as the following the COVID-19 pandemic. These proactive measures are vital for , ensuring that only reach the market.
Notably, the FDA's emphasis on over-the-counter (OTC) products resulted in 37 warning letters targeting compliance issues in categories such as hand sanitizers and sunscreens, highlighting persistent safety concerns. Companies that fail to adhere to a face severe repercussions, as highlighted by recent cases where organizations were cited for significant deficiencies in and data integrity.
The consequences of in a extend beyond regulatory sanctions; they can threaten , as shown by instances where contaminated products infiltrated the market, posing serious risks to consumers. Therefore, organizations must remain vigilant in their and contribute to the overall safety of the pharmaceutical landscape.

Learning CGMP: Resources for New Drug Companies
New drug companies have access to a wealth of resources to deepen their understanding of the . Key among these are , which outline and expectations. Interacting with these documents enables organizations to grasp the fundamental concepts of the , ensuring they can develop effective adherence strategies from the outset. Importantly, the FDA emphasizes the adaptability producers possess in determining 'significant phases' in the manufacturing process, which can aid in customizing to specific operational requirements.
In addition to FDA resources, industry workshops and online play a crucial role in educating teams about the requirements. Successful often incorporate and interactive elements, enhancing both the learning experience and retention of critical information. For instance, programs that focus on practical applications within the have shown a significant improvement in adherence rates among participants. that empower organizations to implement these best practices effectively.
Expert opinions underscore that in a rapidly evolving regulatory landscape. The FDA advocates for a to in-process controls and testing, which is essential for establishing robust regulatory frameworks. By , new pharmaceutical firms can build a solid foundation within a , ultimately leading to improved product standards and regulatory success. Furthermore, manufacturers must substantiate their in-process controls scientifically, ensuring alignment with regulatory expectations.
Training Programs: Essential for CGMP Compliance
Implementing is essential for ensuring throughout all levels of an organization. These programs must address key topics such as:
- Excellence control
- , including the critical stages of (CSV)
Understanding the CSV process—planning, defining user requirements, design specifications, building and configuring systems, and conducting installation, operational, and performance qualifications—ensures that employees are well-versed in . Regular training sessions reinforce the significance of adherence and keep employees informed about any changes in regulations. This approach ultimately enhances and strengthens the organization's commitment to .

Evolving CGMP Regulations: Staying Updated for Compliance
are continually evolving as the FDA updates its guidelines and industry standards advance. Organizations face that necessitate the implementation of robust processes for tracking and adjusting practices accordingly. Engaging with industry newsletters, attending relevant conferences, and joining professional organizations are effective strategies for staying abreast of the latest developments within the .
For instance, organizations that have adopted have reported a 23% decrease in waste and enhanced adherence rates. Furthermore, entities excelling in adherence often achieve 98-99% batch right-first-time rates, underscoring the importance of remaining knowledgeable. As regulatory scrutiny intensifies, proactive oversight of good manufacturing practices in a becomes not only advantageous but crucial for preserving and maintaining a competitive edge.
Key Takeaways: Essential CGMP Facts for Compliance Officers
Oversight officers must prioritize the following essential takeaways:
- is fundamental for ensuring in a cgmp environment, serving as a cornerstone of .
- Non-adherence can result in significant repercussions, including .
- are critical, with competency rates improving to 94% after practical verification.
- Staying informed about regulatory changes is vital, as 72% of pharmaceutical quality professionals report challenges in keeping pace with evolving requirements.
- for demonstrating adherence, as it helps prevent deviations from established protocols and supports organizational credibility. In fact, 40% of FDA warning letters cite data integrity violations, underscoring the necessity of meticulous record-keeping.
By adhering to these principles, compliance officers can adeptly navigate the complexities of the cgmp environment, ensuring not only compliance but also fostering a culture of quality and safety within their organizations.

Conclusion
Navigating the complexities of the Current Good Manufacturing Practices (CGMP) environment is crucial for organizations aiming to ensure product safety and quality in the pharmaceutical and biotechnology sectors. The insights shared highlight the importance of robust compliance strategies, as well as the evolving landscape of regulatory requirements that companies must adeptly manage to safeguard public health and maintain their market reputation.
Key arguments presented in the article underscore the necessity of adhering to CGMP standards, illustrated through real-world examples of successful compliance and the dire consequences of non-adherence. The emphasis on data integrity, comprehensive training programs, and continuous monitoring of regulatory updates are pivotal for organizations striving to achieve excellence in manufacturing practices. Moreover, the FDA's proactive measures against CGMP violations serve as a reminder of the critical nature of compliance in protecting consumers and ensuring the integrity of the pharmaceutical supply chain.
Ultimately, the commitment to a CGMP environment transcends regulatory obligation; it is a moral imperative that fosters consumer trust and enhances operational efficiency. Organizations are encouraged to prioritize ongoing education, embrace best practices, and remain vigilant in their compliance efforts. By doing so, they not only mitigate risks but also position themselves as leaders in a competitive landscape, ultimately contributing to the advancement of public health and safety.
Frequently Asked Questions
What are Current Good Manufacturing Practices (CGMP)?
Current Good Manufacturing Practices (CGMP) are regulations enforced by the FDA to ensure that manufacturing processes and facilities are designed, monitored, and controlled effectively to guarantee product safety and quality.
Why are CGMP important in the pharmaceutical industry?
CGMP are vital for ensuring that pharmaceutical products are safe, effective, and meet high standards, thereby mitigating risks of contamination, mix-ups, and manufacturing errors, which protects consumer health and enhances company credibility.
What are the core principles of CGMP?
The core principles of CGMP include maintaining clean and hygienic environments, ensuring consistent production and quality control, and implementing robust documentation practices for traceability and regulatory verification.
Can you provide an example of successful CGMP compliance?
AVS Life Sciences assisted a pharmaceutical manufacturer in enhancing their facility from a Biosafety Level 1 to a Level 2 GMP environment for lentivirus vector material production, demonstrating adherence to FDA regulations and GXP standards through meticulous documentation and validation processes.
What are the consequences of failing to adhere to CGMP?
Failure to comply with CGMP can lead to significant consequences, including costly product recalls and FDA-issued warning letters, highlighting the necessity for strict adherence to these practices.
How does the FDA monitor compliance with CGMP?
The FDA monitors compliance through inspections and documentation reviews, as indicated by the issuance of Form 483s that document deficiencies found during inspections.
What current trends are influencing CGMP practices?
There is a growing emphasis on data integrity and management systems, with initiatives like the FDA's New Inspection Protocol Project (NIPP) aiming to improve inspection efficacy through a targeted, data-driven approach.
How can organizations benefit from implementing CGMP?
Organizations that commit to CGMP can improve operational efficiency, enhance product integrity, safeguard public health, and bolster their market reputation, leading to greater success in a competitive landscape.
List of Sources
- AVS Life Sciences: Key Insights on Current Good Manufacturing Practices (CGMP)
- firsteigen.com (https://firsteigen.com/blog/data-quality-issues-affecting-the-pharmaceutical-industry-finding-a-solution)
- Understanding Current Good Manufacturing Practices in Pharma (https://avslifesciences.com/blog-post/understanding-current-good-manufacturing-practices-in-pharma)
- gmp-compliance.org (https://gmp-compliance.org/gmp-news/pharmaceutical-industry-fda-data-on-the-level-of-quality)
- fdli.org (https://fdli.org/2021/05/pharmaceutical-gmps-quality-control-and-data-a-deeper-look-at-fdas-fy-2020-fda-observations)
- Understanding CGMP: Definition and Core Principles
- FDA Issues New Guidance on Current Good Manufacturing Practices for Drugs | Advisories | Arnold & Porter (https://arnoldporter.com/en/perspectives/advisories/2025/01/fda-guidance-good-manufacturing-practices-for-drugs)
- Complete Guide to Current Good Manufacturing Practices (cGMP) (https://cgmpconsulting.com/complete-guide-to-cgmp)
- gmpplastic.com (https://gmpplastic.com/blogs/useful-articles-on-lab-supplies-faq-section/info-about-the-current-good-manufacturing-practice-cgmp?srsltid=AfmBOooW0B9jd2xfeV98usdbrx7W1m4wp12Wy4QvyZ-yBOc_8iam5-Bb)
- Facts About the Current Good Manufacturing Practices (CGMP) (https://fda.gov/drugs/pharmaceutical-quality-resources/facts-about-current-good-manufacturing-practice-cgmp)
- Importance of CGMP: Ensuring Product Safety and Quality
- cfpie.com (https://cfpie.com/the-importance-of-cgmp-in-pharma-supply-chain-industry)
- pharmamanufacturing.com (https://pharmamanufacturing.com/compliance/cgmps/article/11358460/pharma-manufacturing-gmps-cgmps-and-statistics-pharmaceutical-manufacturing)
- Understanding the Importance of the FDA’s Current Good Manufacturing Practices - Medical Solutions by UFP MedTech (https://ufpmedtech.com/article/understanding-the-importance-of-the-fdas-current-good-manufacturing-practices)
- pjrfsi.com (https://pjrfsi.com/the-five-whys-unlocking-the-importance-of-current-good-manufacturing-practices-cgmp)
- FDA Compliance Assessment: How the FDA Evaluates CGMP Adherence
- FDA Issues New Guidance on Current Good Manufacturing Practices for Drugs | Advisories | Arnold & Porter (https://arnoldporter.com/en/perspectives/advisories/2025/01/fda-guidance-good-manufacturing-practices-for-drugs)
- Yogi Berra's Non-sensical Quotes on Good Manufacturing Practices (cGMP) (https://linkedin.com/pulse/yogi-berras-non-sensical-quotes-good-manufacturing-practices-ahn)
- datadashboard.fda.gov (https://datadashboard.fda.gov/oii/cd/inspections.htm)
- americanpharmaceuticalreview.com (https://americanpharmaceuticalreview.com/Featured-Articles/587071-A-Discourse-on-Pharmaceutical-cGMP-FDA-Form-483-Trends-Why-are-We-Re-Living-the-Same-Issues-Over-the-Last-23-Years)
- descase.com (https://descase.com/case_studies/storage-handling-case-study)
- Consequences of Non-Compliance: Risks to Drug Safety
- lightfootlawdc.com (https://lightfootlawdc.com/blogs/fda-drug-recall-statistics)
- europeanpharmaceuticalreview.com (https://europeanpharmaceuticalreview.com/news/184193/fda-releases-analysis-on-drug-product-quality-in-2022)
- 10 inspirational quotes for the pharma sector (https://pharmaceuticalmanufacturer.media/pharmaceutical-industry-insights/10-inspirational-quotes-for-the-pharma-sector)
- FDA Actions Against CGMP Violations: Protecting Public Health
- pubmed.ncbi.nlm.nih.gov (https://pubmed.ncbi.nlm.nih.gov/30227064)
- researchgate.net (https://researchgate.net/publication/384046692_FDA_Warning_Letter_Trends_A_15-Year_Analysis)
- outsourcedpharma.com (https://outsourcedpharma.com/doc/trends-in-fda-fy2023-inspection-based-warning-letters-0001)
- insider.thefdagroup.com (https://insider.thefdagroup.com/p/fake-data-failed-tests-and-more-four)
- Learning CGMP: Resources for New Drug Companies
- FDA Issues New Guidance on Current Good Manufacturing Practices for Drugs | Advisories | Arnold & Porter (https://arnoldporter.com/en/perspectives/advisories/2025/01/fda-guidance-good-manufacturing-practices-for-drugs)
- Yogi Berra's Non-sensical Quotes on Good Manufacturing Practices (cGMP) (https://linkedin.com/pulse/yogi-berras-non-sensical-quotes-good-manufacturing-practices-ahn)
- 10 inspirational quotes for the pharma sector (https://pharmaceuticalmanufacturer.media/pharmaceutical-industry-insights/10-inspirational-quotes-for-the-pharma-sector)
- researchgate.net (https://researchgate.net/publication/265024648_Statistics_in_Pharmaceutical_Development_and_Manufacturing)
- udemy.com (https://udemy.com/course/statistical-evaluation-of-stability-study-in-pharmaceuticals?srsltid=AfmBOoorPvmr-vnysblJfMXc4feangx5Af4ZKwIXAgZ7KDKZsxt0a4U5)
- Training Programs: Essential for CGMP Compliance
- Facts About the Current Good Manufacturing Practices (CGMP) (https://fda.gov/drugs/pharmaceutical-quality-resources/facts-about-current-good-manufacturing-practice-cgmp)
- When Training Falls Short: FDA Findings on GMP Training Deficiencies in 2025 - ECA Academy (https://gmp-compliance.org/gmp-news/when-training-falls-short-fda-findings-on-gmp-training-deficiencies-in-2025)
- The Ultimate List of Compliance Program Statistics (https://navexglobal.com/compliancenext/understanding-the-basics/the-ultimate-list-of-compliance-program-statistics)
- 5 Important Things for Life Sciences Companies to Consider | MasterControl (https://mastercontrol.com/gxp-lifeline/quality-inspiration)
- Key Metrics for GMP Training in Pharmaceutical Systems: Leading & Lagging Indicators (https://investigationsquality.com/2025/02/04/key-metrics-for-gmp-training-in-pharmaceutical-systems-leading-lagging-indicators)
- Evolving CGMP Regulations: Staying Updated for Compliance
- westbourneit.com (https://westbourneit.com/6-regulatory-trends-in-the-pharmaceutical-industry-to-be-aware-of-in-2024)
- Navigating the Complex World of Pharma Regulatory Compliance: Finding the Sweet Spot Between Safety & Innovation | GMP Pros (https://gmppros.com/pharma-regulatory-compliance)
- 10 inspirational quotes for the pharma sector (https://pharmaceuticalmanufacturer.media/pharmaceutical-industry-insights/10-inspirational-quotes-for-the-pharma-sector)
- thefdagroup.com (https://thefdagroup.com/blog/2019-fda-warning-letter-inspection-observation-trends)
- Yogi Berra's Non-sensical Quotes on Good Manufacturing Practices (cGMP) (https://linkedin.com/pulse/yogi-berras-non-sensical-quotes-good-manufacturing-practices-ahn)
- Key Takeaways: Essential CGMP Facts for Compliance Officers
- Importance of Documentation - Expert and Influential Leader Quotes : Erase Your Risk with Proper Documentation (https://forensicnotes.com/importance-of-documentation-expert-and-influential-leader-quotes)
- Navigating the Complex World of Pharma Regulatory Compliance: Finding the Sweet Spot Between Safety & Innovation | GMP Pros (https://gmppros.com/pharma-regulatory-compliance)
- pharmamanufacturing.com (https://pharmamanufacturing.com/compliance/cgmps/article/11358460/pharma-manufacturing-gmps-cgmps-and-statistics-pharmaceutical-manufacturing)
- The Crucial Role of Documentation for GMP Compliance - InstantGMP (https://instantgmp.com/improve-gmp-compliance-with-documentation)
